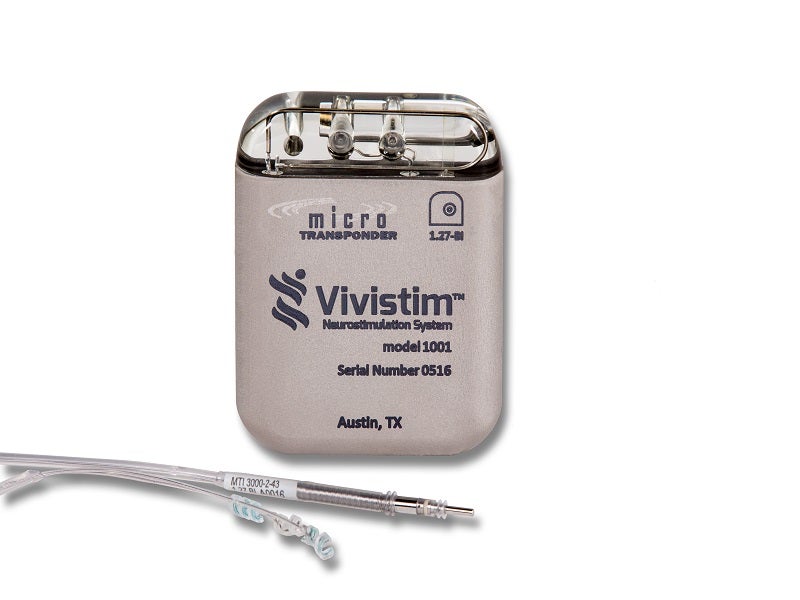
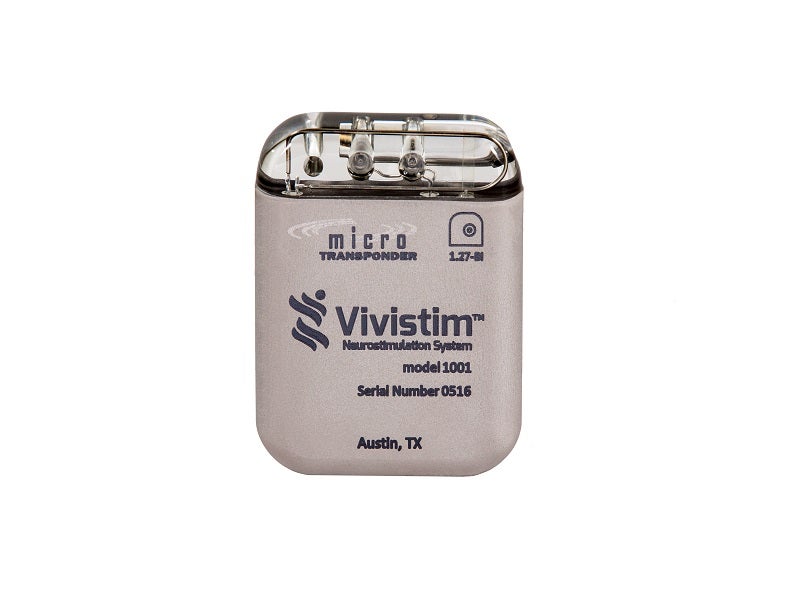
The Vivistim® Paired Vagus Nerve Stimulation (VNS) System is a first-of-its-kind, drug-free implantable neurostimulation device indicated to treat moderate to severe upper limb mobility issues associated with chronic ischaemic stroke alongside post-stroke rehabilitation therapy.
Developed by US-based medical device manufacturer MicroTransponder, the Vivistim system electrically stimulates the vagus nerve to improve the motor commands relayed by the brain to ensure the movement of arms and hands.
The Vivistim Paired VNS system received approval from the US Food and Drug Administration (FDA) under the premarket approval pathway in August 2021. The device also holds breakthrough device designation from the FDA.
Vivistim Paired VNS system design
The Vivistim system comprises four main components, namely an implantable pulse generator (IPG) stimulator, the lead, stroke application and programming software (SAPS), and a wireless transmitter.
The IPG stimulator weighs less than 70g and is implanted on the left vagus nerve, which generates electrical pulses at frequencies of 1Hz to 30Hz.
A 43cm silicone lead wire, with a cuff diameter of 2mm or 3mm, is tunnelled subcutaneously to an area created in the left pectoral region, where it is connected to the IPG stimulator.
The SAPS software is loaded onto a laptop to be used by therapists to generate impulses using a push-button controller during rehabilitative exercises. The wireless transmitter communicates changes made to the IPG settings through software.
Vivistim Paired VNS system mechanism of operation
A physical therapist stimulates VNS for half a second by a push-button remote control while the patient makes certain movements. The stimulator, implanted beneath the skin on the patient’s left vagus nerve in the left carotid sheath, sends a mild electrical pulse generated by IPG.
The implanted lead wire sends electrical stimulation up to the brain. The electrical signal travels to two separate brain areas via the nucleus tractus solitarius.
Brain chemicals known as neuromodulators are released from the areas of the brain that receive the electrical signal. Neuromodulators are also essential for learning and memory.
The transmitter release increases the relevance of the physical therapy. The resulting brain reorganisation and strengthening of neural links improve motor function in the brain.
Rehabilitation sessions take place three times a week for six weeks. Each session lasts for 75 to 90 minutes, during which movements and stimulation occur concurrently every five to ten seconds.
Patients are also given a magnet to use at home to stimulate the nerve for 30 minutes a day during stimulation sessions.
Benefits of the neurostimulation device
The Vivistim system has been clinically proven to restore two to three times more arm and hand function compared to rehabilitation alone after six weeks of in-clinic therapy.
The drug-free rehabilitation system is extremely efficient and around 98% of patients reported they were satisfied with their treatment using the device.
The system can be used in both clinical and at-home settings, as well as post-stroke rehabilitation treatment.
Clinical trials on the Vivistim Paired VNS system
The Vivistim Paired VNS system’s safety and effectiveness were evaluated in a pivotal clinical trial, in which 108 patients were enrolled.
The clinical study was conducted at 19 clinical sites across the US and UK. Patients were divided into two groups, namely a study group (53 patients) and a control group (55 patients). Both groups completed 300 to 400 physical activities for 90 minutes a day, three times a week for six weeks.
The control group received extremely low levels of VNS stimulation for the first five exercises and had no stimulation for the remaining session. The treatment group received adequate VNS stimulations for all the sessions.
Physical therapy sessions were organised for both groups in an equal quantity and quality. The patients were given follow-up assessments on the first, 30th and 90th days after the initial six-week study.
The Upper Extremity Fugl-Meyer Assessment (FMA-UE), a stroke-specific assessment of motor dysfunction, was used to assess the effectiveness. Progress was assessed as an increase in motor activity from baseline after six weeks of treatment.
Patients in the treatment group had an average score increase of five points, whereas the control group’s score increased by 2.4 points on average.
Around 47.2% of patients in the treatment group had an average score increase of six or more points on the FMA-UE score 90 days after therapy, compared to 23.6% in the control group.
Adverse events observed with the Vivistim system during the clinical study were bruising, general hoarseness and pain, low mood, fracture, rash, throat irritation, difficulty in speaking, falling, hoarseness after surgery, muscle pain, headache, fatigue, urinary tract infection and dizziness.