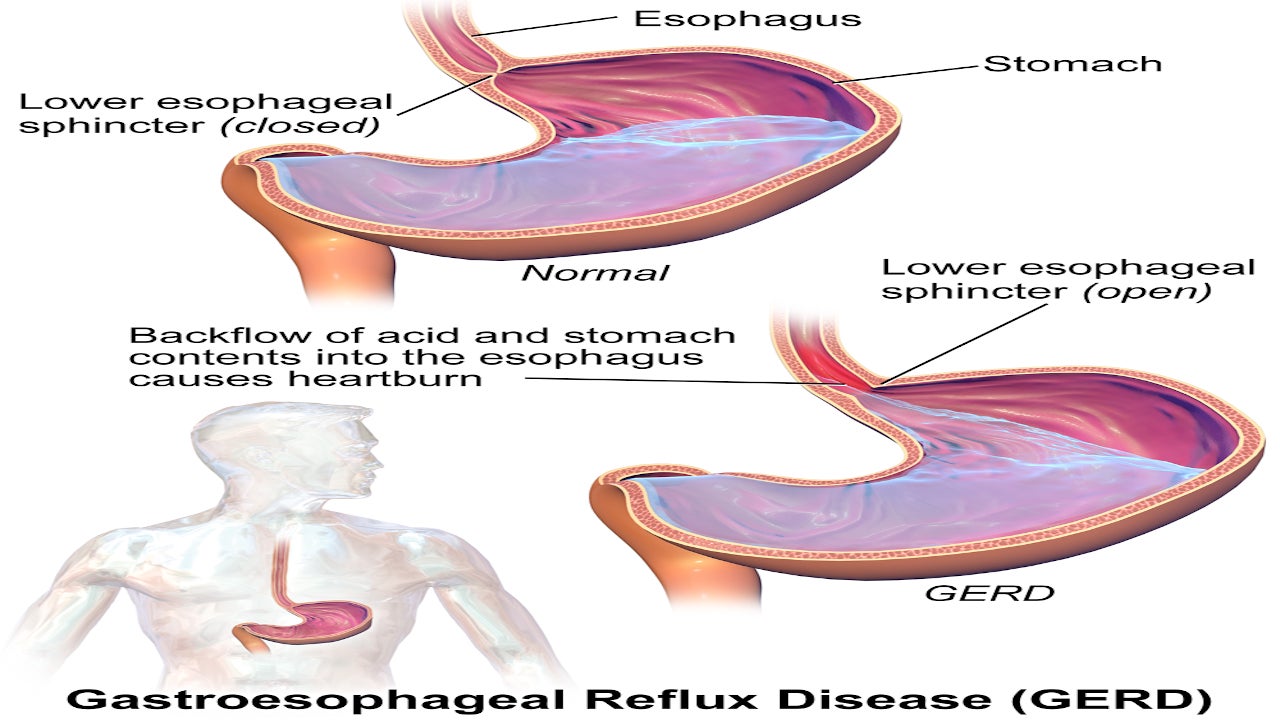
MiVu™ is the first mucosal integrity (MI) testing system for rapid diagnosis of gastroesophageal reflux disease (GERD), eosinophilic oesophagitis (EoE) and Non-GERD, as well as monitor treatment response in patients with GERD and EoE.
Developed by Diversatek Healthcare in collaboration with Vanderbilt University, MiVu detects oesophageal mucosal changes during a routine endoscopy, indicating the presence or likelihood of GERD and other oesophageal conditions in two minutes.
In December 2020, the US Food and Drug Administration (FDA) cleared the testing system with De Novo classification due to its safety and efficacy profile in obtaining electrical properties measurements within the oesophageal tissue.
MiVu testing system details
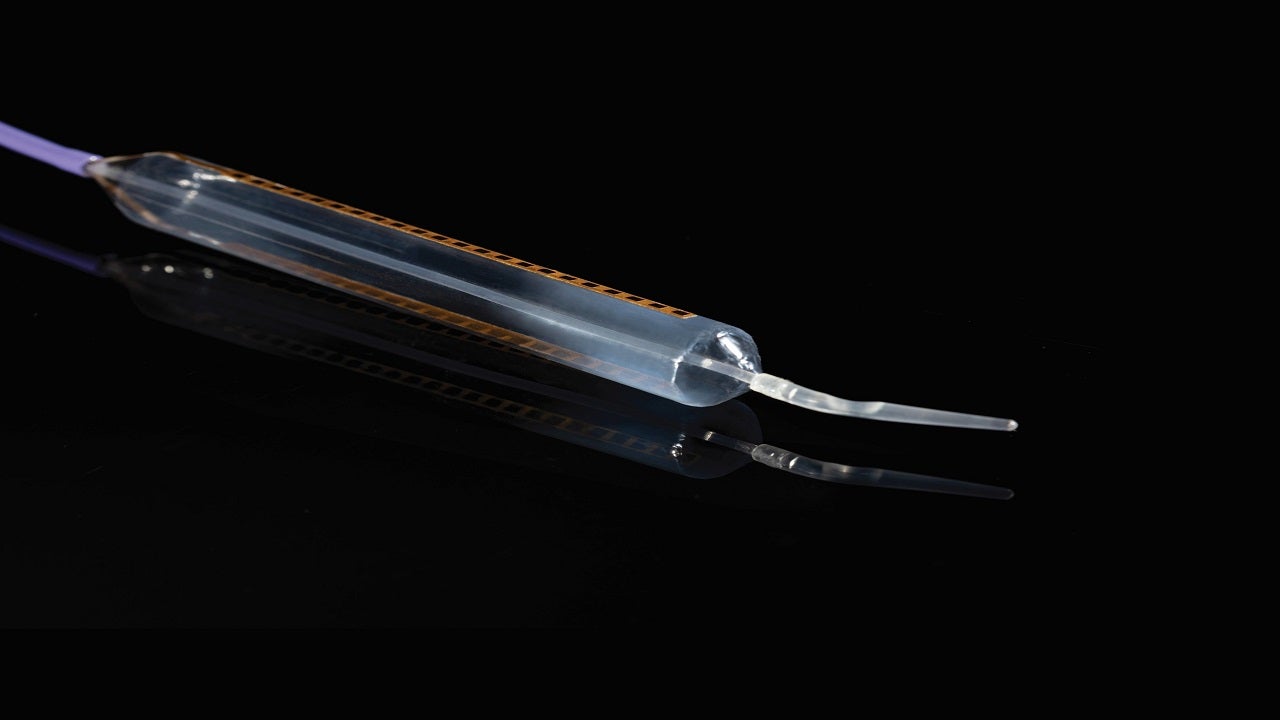
MiVu uses a balloon probe and proprietary software to measure oesophageal epithelium conductivity directly and provides a mucosal integrity contour pattern and disease probability to clinicians.
The MiVu balloon probe of the system is a single-use, disposable supply that meets the requirements of the CMS as a separately reportable supply device. It incorporates both radial and axial impedance sensors mounted at 180° intervals in a 10cm section of the oesophagus that evaluate mucosal integrity and provide real-time impedance values, reducing variability in measurement.
Real-time detection via MiVu during a routine endoscopy potentially eliminates the need for 24 hours to 48 hours ambulatory pH or histopathological oesophageal biopsies to reduce both diagnosis and treatment latency.
Limitations with current testing methods
Key diagnostic techniques currently used for suspected GERD are endoscopy and ambulatory reflux monitoring, which are widely used but have major drawbacks that result in an ambiguous and questionable diagnosis inaccurate in determining therapy for diagnosed patients.
Oesophageal mucosal inflammation detected by endoscopic assessment is present in less than 30% of patients.
Currently, available pH technologies only test the acidity of the refluxed substance at a single point in the oesophagus. Additionally, these tests do not determine day-to-day reflux variability and only include 24 hours to 48 hours snapshot of a chronic disease process.
Thus, current suboptimal diagnostic testing in GERD can have approximately 30% of false-negative rates in patients with endoscopic oesophagitis.
Mucosal integrity (MI) testing
The presence of dilated intercellular spaces (DIS) or spongiosis impacts the paracellular permeability of the oesophageal lumen, affecting mucosal integrity.
DIS is a vital histological feature in GERD and EoE where the degree of dilation inversely corresponds with MI measurements and, therefore, lower impedance values show increasing DIS.
Oesophageal mucosal integrity assessments will allow clinicians to distinguish certain oesophageal diseases and monitor GERD response quickly and efficiently.
In patients with the non-erosive disease, having irregular pH reflux monitoring, MI values were close to those with erosive oesophagitis. MI was better than pH monitoring in predicting erosive reflux, with an accuracy of 95% and a positive predictive value of 96% compared to 64% and 40%, respectively.
Despite the absence of apparent mucosal modifications in endoscopy, MI can distinguish an irregular reflux pattern from usual reflux parameters.
MI also successfully predicted EoE during endoscopy with a 100% sensitivity and a 96% specificity without any histology requirement.
Benefits of MiVu
MiVu reduces the need for ambulatory reflux testing and multiple invasive biopsies. The direct measurement by MiVu offers reliable and instant results that can lead to therapy decisions and monitor the activity of diseases over time, effectively saving time and money for patients.
It minimises the need for unnecessary GI testing and offers objective evidence that there is no inflammatory disorder.