Navitor™ is a next-generation transcatheter aortic valve implantation (TAVI) system developed by Abbott, a medical device company based in the US, to treat people with severe aortic stenosis, a common and life-threatening heart valve disease.
It is an addition to Abbott’s comprehensive structural heart transcatheter portfolio, which includes the Portico™ TAVI system and MitraClip™ transcatheter edge-to-edge repair system and offers less invasive options for the treatment of heart diseases to physicians and patients.
The Navitor TAVI system received the CE Mark in Europe to treat aortic stenosis in May 2021. The device was launched in India in December 2022.
The system was approved by the US Food and Drug Administration (FDA) in January 2023.
In August 2025, Abbott obtained approval in Europe for using the device as a minimally invasive alternative to surgery in patients with symptomatic, severe aortic stenosis who are classified as low or intermediate surgical risk.
Navitor TAVI system design and features
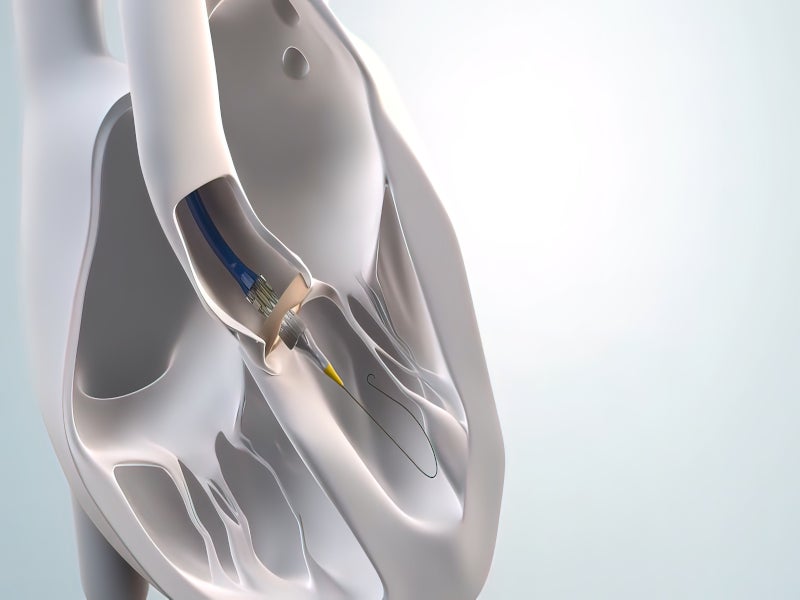
The Navitor TAVI system is indicated for transcatheter delivery in patients with symptomatic severe native aortic stenosis. It is a minimally invasive substitute for surgery for aortic stenosis patients who are at high or extremely high risk for open-heart surgery.
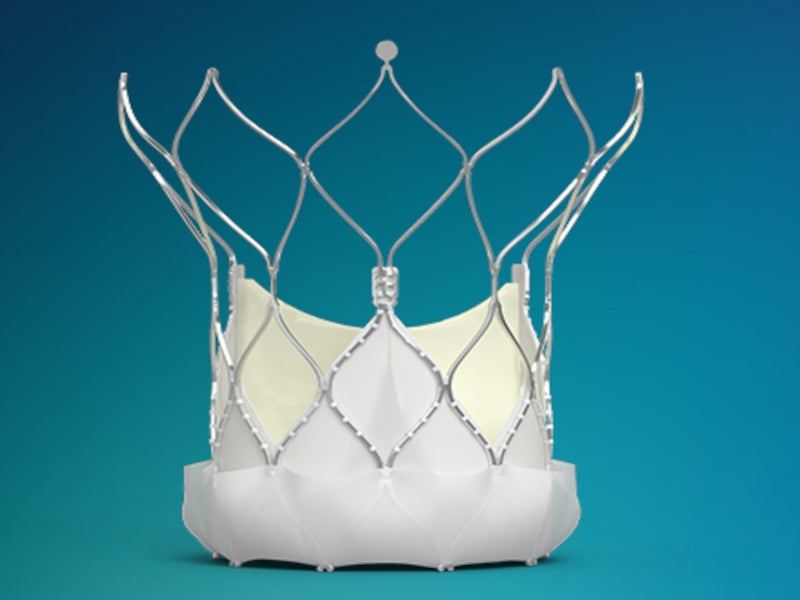
The system is designed for immediate functionality, continuous stability and no rapid pacing. It comprises the Navitor valve and FlexNav™ delivery system. The valve comes in 23mm, 25mm, 26mm, 27mm and 29mm sizes.
A 29mm Navitor valve features a 21F equivalent integrated sheath diameter. It has 36 cells in total, of which nine cells are in the annulus section of the stent.
Navitor valve details
The Navitor valve features a curved aortic stent with NaviSeal™ Cuff smart paravalvular leak (PVL) sealing technology, which actively synchronises with the cardiac cycle. Intra-annular leaflets within the native valve help to seal and manage PVL by self-expanding, filling the gaps between the annulus and the valve caused by calcification.
The exclusive Linx™ anti-calcification technology of the valve extends its performance by preventing calcification in four different ways.
Its intelligent design offers optimised radial force, improving expansion, anchoring, stability and sealing across all sizes. Large frame cells and intra-annular valve design help to preserve access to coronary vessels for future intervention.
The atraumatic aortic stent cells of the valve reduce the risk of injury to biological structures. Tissue ingrowth and healing are completed within three months of valve implantation.
FlexNav delivery system details
FlexNav facilitates the implantation of the Navitor valve in blood vessels with a diameter of equal to or more than 5mm. The delivery system has a working length of 107cm.
It has a recapturable, repositionable and retrievable design with 14F and 15F equivalent integrated sheath diameters. The working length of the integrated sheath is 30cm.
The low-profile and highly flexible catheter provides excellent deliverability by allowing easy access to small vessels and tortuous patient anatomies.
It features a flexible capsule with a hydrophilic coating, which reduces friction and allows it to glide smoothly through the vasculature. The atraumatic nosecone of the FlexNav reduces the risk of vascular complications and calcium dislodgement, while the stability layer offers controlled deployment and accurate valve placement.
Haemodynamics
The self-expanding valve and non-tapered stent of the Navitor TAVI system offer haemodynamic stability for a calm and controlled deployment.
The large effective orifice areas along with the non-tapered stent result in single-digit gradients, leading to improved cardiac function, long-term durability and minimal prosthesis-patient mismatch.
Clinical studies on Navitor TAVI system
The Portico NG (Next Generation) study, a prospective, multi-centre, international, single-arm trial, underpinned the FDA approval of the Navitor TAVI system. The study enrolled 333 patients with symptomatic, severe native aortic stenosis.
Results from Portico NG indicated that Navitor is a safe and effective treatment option for patients with severe, symptomatic aortic stenosis who are at high or greater surgical risk.
Navitor demonstrated a 97% procedural success rate, while the incidence of all-cause mortality was 1.9%, and the rate of disabling stroke was also 1.9%. PVL outcomes were strong, with 0% moderate-or-greater PVL at 30 days, supporting the effectiveness of the NaviSeal active sealing cuff.
The CE Mark approval of the Navitor TAVI device in Europe was based on results from the VANTAGE open-label, single-arm interventional clinical study.
A total of 434 patients were enrolled for the study, who underwent transcatheter aortic valve replacement with the Navitor valve and FlexNav delivery system.
The primary endpoint of the study was the combined all-cause mortality or disabling stroke in a time frame of 12 months.
Among the first 262 patients who completed 12‑month follow‑up, the combined incidence of all‑cause mortality or disabling stroke was 2.3%.
At 30 days, no patients experienced moderate or greater PVL and 13.6% had mild PVL, a proportion described as low.
The procedure achieved a technical success rate of 97%, with no deaths occurring during the intervention, and haemodynamic performance remained favourable at 12 months.