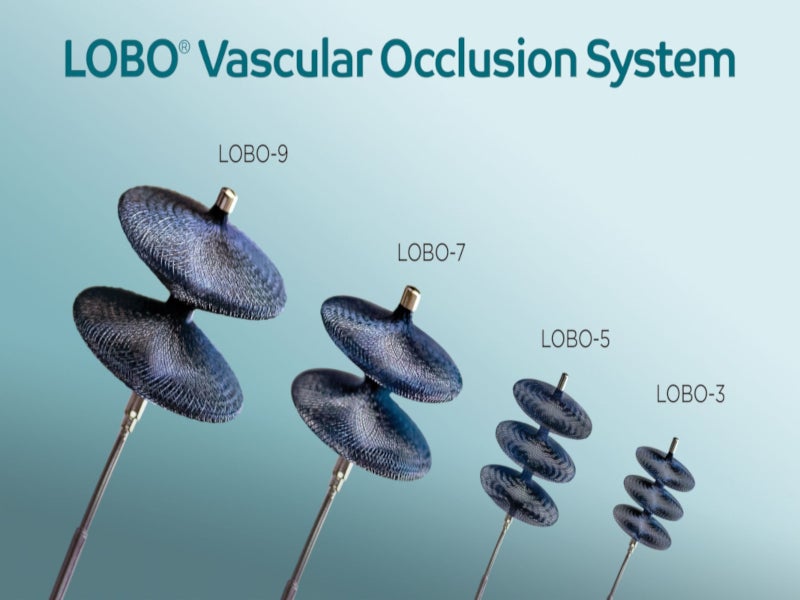
Low-profile Braided Occluder (LOBO®) is a purpose-built, microcatheter-delivered device including the LOBO-3, LOBO-5, LOBO-7 and LOBO-9 vascular occluders developed by Okami Medical for fast and complete occlusion of various peripheral blood vessels.
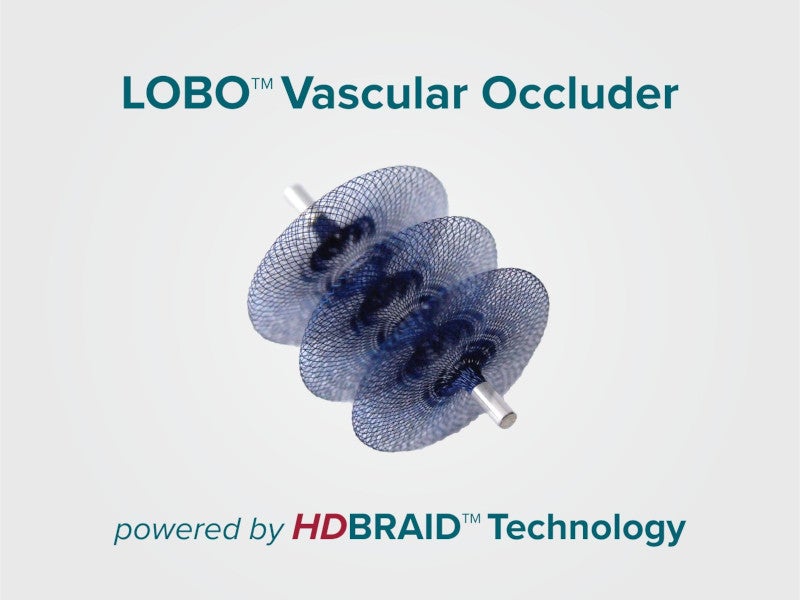
LOBO® vascular occluders are the first peripheral embolisation device that is powered by proprietary HDBRAIDTM technology, an innovative braiding method for developing vascular occlusion devices.
The LOBO® vascular occlusion system received 510(k) clearance from the US Food and Drug Administration (FDA) in November 2019. LOBO-5 vascular occluder, the company’s second offering in the LOBO® vascular occlusion system portfolio, received the FDA 510(k) clearance in December 2020, while LOBO-7 and LOBO-9 vascular occluders were cleared by the FDA in June 2022.
The LOBO® occluder devices provide interventional physicians with a minimally invasive single medical device solution for the occlusion of various arterial targets, even in small and inaccessible locations. It helps in the efficient closure of peripheral vessels throughout the body for the treatment of different conditions.
Design and features
The LOBO® vascular occlusion system integrates a patented design with HDBRAID™ technology to form a highly occlusive pore structure that quickens the vessel closure by significantly reducing the blood flow. The Lobo-3 occluder is intended for use in blood vessels ranging from 1.5mm to 3mm in diameter. LOBO-5 is intended for use in 3mm to 5mm diameter vessels, whereas LOBO-7 and LOBO-9 can be used in 5mm to 7mm and 7mm to 9mm diameter blood vessels, respectively.
The LOBO® occluder features a three-disc design that intensifies the occlusive effects of HDBRAIDTM and improves its trackability to small and twisted vessels. It can expand the accessible anatomical locations and conforms to the curved vessels.
The intuitive LOBO® occlusion system has user-friendly features such as compatibility with low-profile 0.027in microcatheter, radiopaque marker bands for visualisation, full retrievability and state-of-the-art, instantaneous mechanical detachment.
LOBO vascular occlusion system working
A suitable LOBO® occluder is advanced through the delivery catheter into a blood vessel and positioned for deployment during a process. The occluder starts self-expanding to securely engage the vessel wall, as soon as the delivery catheter is retracted, creating a highly stable implant. The flexible occluder conforms to a wide range of targets, including highly curved vessels.
The retraction of a button located on the easy-to-use mechanical detachment handpiece attached to the pusher wire allows the instant release of the LOBO® occluder and retraction of the pusher wire. The dense pore structure of the system enables the expansion of vessel lumen that rapidly blocks blood flow for a fast and efficient occlusion.
The non-clinical testing and MRI simulations of the system have demonstrated that the device is MR conditional and the person with the implant can safely go for MRI scanning if certain magnetic field conditions are followed, which are 1.5T or 3T field strengths and 2000 gauss/cm maximum spatial gradient magnetic field.
HDBRAID technology
HDBRAIDTM technology is an innovative braiding method that offers four times greater pore density and smaller pores than the standard braiding methods. The pore structure helps significantly reduce the blood flow to a target location, thereby accelerating haemostasis and thrombosis.
It offers more than 5,000 pores for fast and durable occlusion with a single device. The technology allows the system to be highly stable after placement without any spinnaker effect.
LOBO vascular occlusion system benefits
The highly occlusive braided structure of the LOBO® vascular system eliminates the need for having different embolic devices by reducing blood flow and enabling vascular occlusions throughout the body.
Unlike coils with irregular mass and large pores, LOBO® vascular occlusion system offers a high-density and small pore structure for vessel occlusion. It improves procedure stability and helps in case of trauma and unstable bleeding.