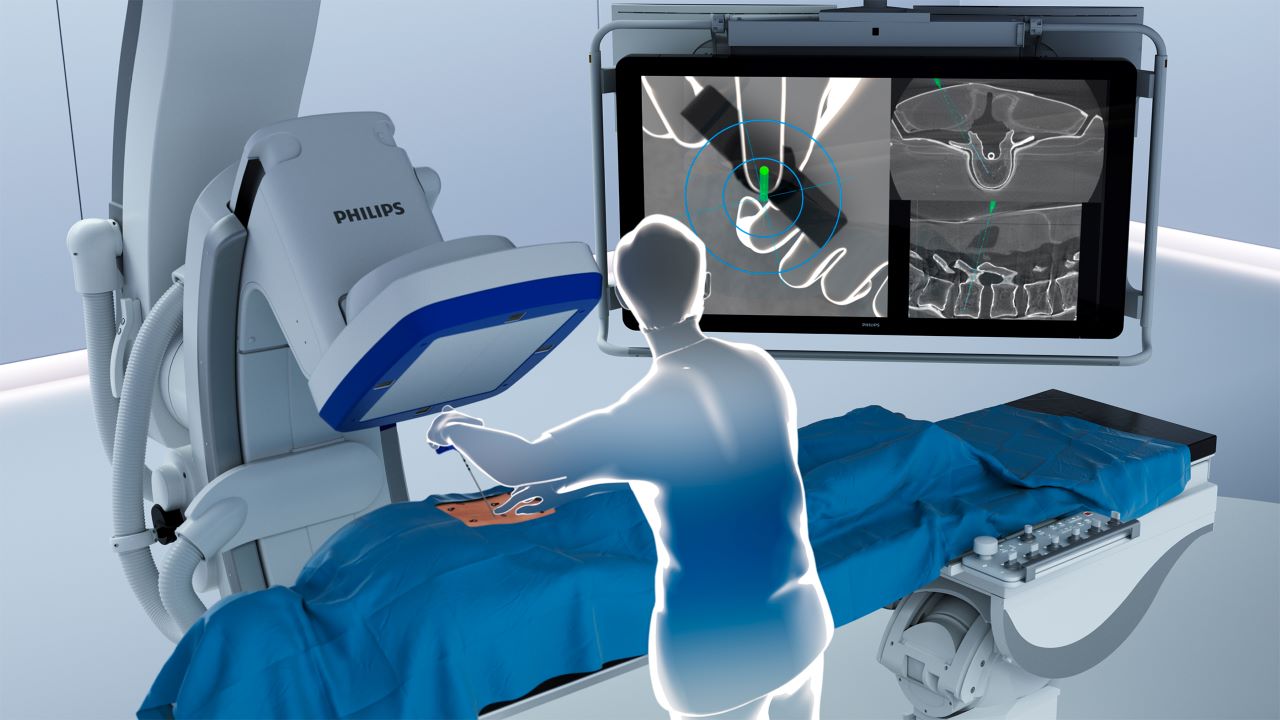
ClarifEye is an augmented reality (AR) surgical navigation system developed by Netherlands-based health technology solutions company Philips to assist minimally invasive spine surgeries in a hybrid operating room.
ClarifEye combines superior 2D and 3D visualisations at a low X-ray dose with 3D AR to give surgeons precise imaging guidance. The solution is integrated with Philips’ Azurion image-guided therapy platform to give live intra-operative visual feedback for accurate screw placement.
Claimed to be Philips’ first AR surgical navigation technology for minimally invasive spine surgery, the company announced the development in January 2017 and launched the system in February 2021.
ClarifEye has been granted the CE mark approval, with 510(k) approval pending with the US Food and Drug Administration (FDA).
Demand for the ClarifEye AR system
Spine conditions affect patients’ quality of life and severe cases may stop them from walking or even moving from their beds.
Minimally invasive spine surgery reduces patients’ post-operative pain, hospital stays, blood loss, soft tissue damage and scar tissue.
Spinal fusion procedures are complex and delicate, involving permanently connecting two or more vertebrae to improve stability, alleviate pain and correct deformities.
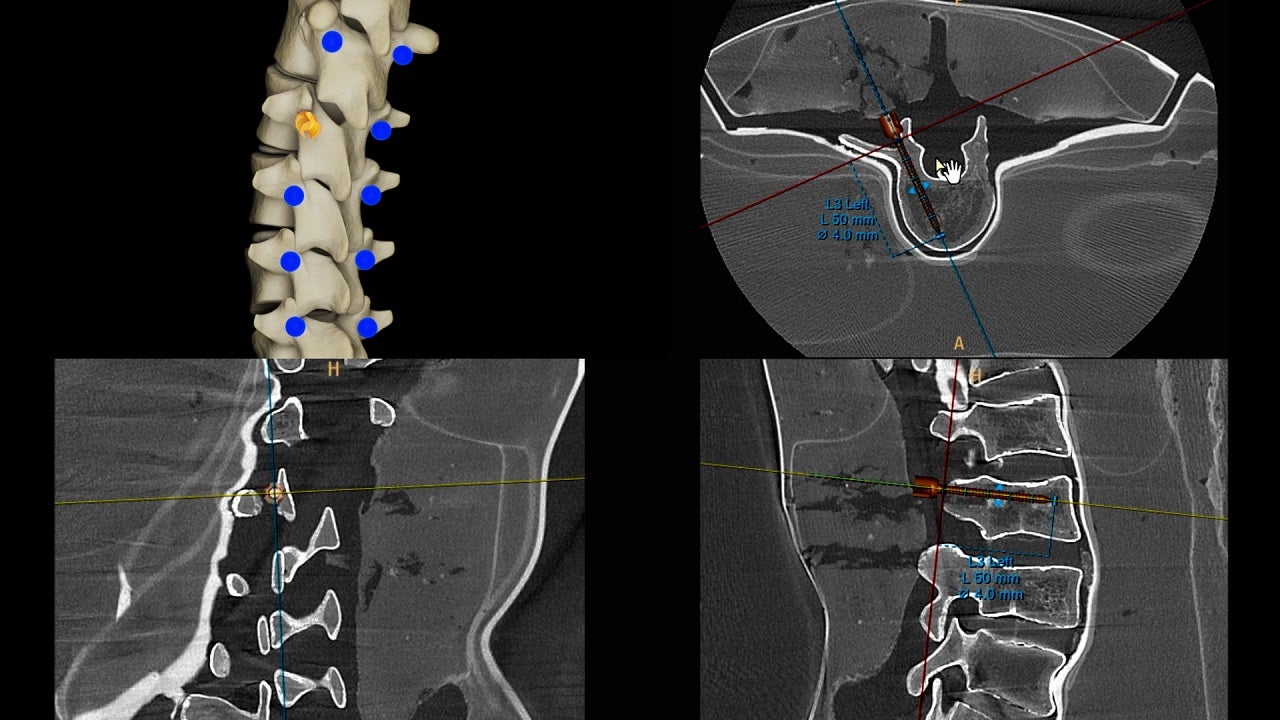
The accurate placement of pedicle screws is a difficult part of minimally invasive procedures. ClarifEye lets surgeons view a 3D image of the patient’s spinal anatomy in order to place screws accurately.
Functionality of the ClarifEye AR solution
ClarifEye works by placing non-invasive markers on the patient’s skin, which are detected by four high-resolution cameras. These cameras video track the markers and visualise the tip of the ClarifEye needle as it follows the planned path in the spine. This boosts the surgical field with 3D cone-beam computed tomography (CT) imaging, removing the need for an additional X-ray.
The system combines both imaging and AR navigation to ensure the precise placement of pedicle screws. By integrating the view of the surgical field with the internal 3D view of the patient, it constructs a 3D AR view of the patient’s external and internal anatomy.
Unlike conventional surgical navigation systems, ClarifEye improves surgical workflow through clinical accuracy and reduces revision surgery rates and post-operative CT scans.
Features of the ClarifEye AR solution
Some key benefits of ClarifEye include its user-friendly interface, excellent image quality and precision, and minimal surgical procedure time.
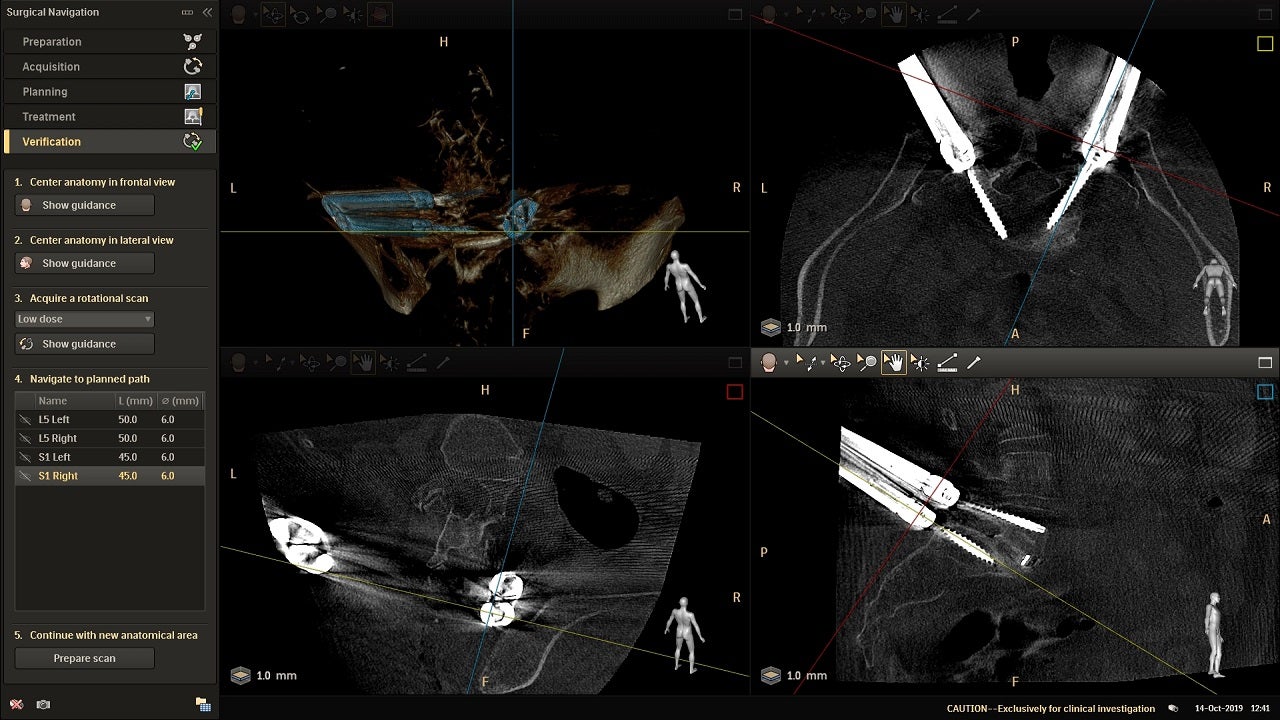
By conducting an intraoperative scan, ClarifEye offers intraoperative image guidance to create a high-resolution 3D model with automatic spine segmentation to aid the precision planning and navigation of devices.
ClarifEye uses high-quality intraoperative cone-beam CT imaging technology to show the complexity of spinal anatomy. The system minimises cone-beam CT radiation doses during spine surgery and allows small and narrow pedicles to be shown.
In its 3D mode, the system allows the position, length and width of the virtual pedicles screw to be adjusted so that surgical procedures can be planned with increased ease and accuracy.
The ClarifEye skin markers also offer non-invasive patient tracking during navigated procedures to streamline the surgical workflow. This allows for intraoperative verification to assess screw placement before the wound is closed.
The system also features live AR needle guidance to offer insightful visualisation of anatomical structures and assist precision without the need for an X-ray.
Clinical studies on the ClarifEye AR solution
A matched control study at the Karolinska University Hospital in Sweden compared pedicle screw placement accuracy and clinical aspects between ClarifEye AR surgical navigation and the conventional fluoro-guided approach.
The study, which involved 20 patients, found that the clinical accuracy of pedicle screw placement with ClarifEye was significantly higher (94%) compared with the conventional fluoro-guided approach (89.6%). The accuracy of screw placement was evaluated using the Gertzbein scale.
In 2020, a usability evaluation for ClarifEye was performed on 14 clinical users in a simulated lab condition in a test lab in Cleveland, Ohio.
Results obtained from the usability evaluation showed that ClarifEye scored 83 on the System Usability Score (SUS) scale.