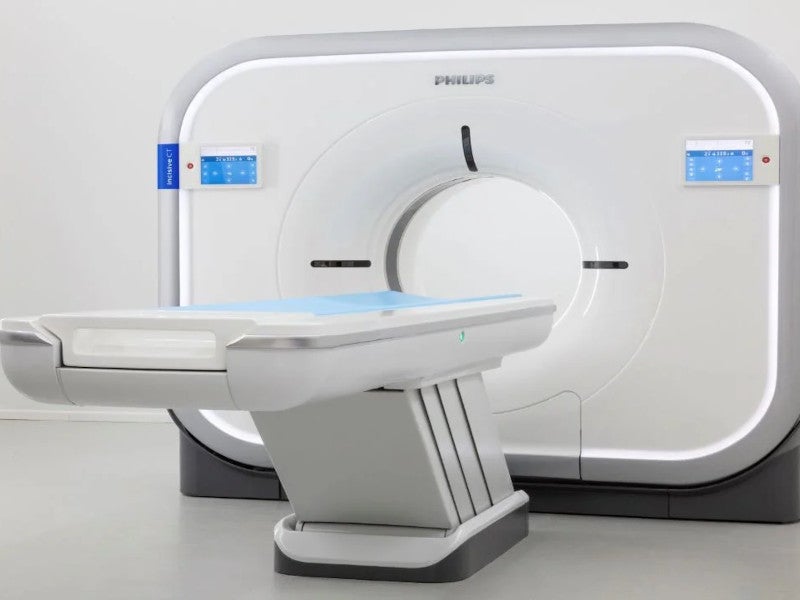
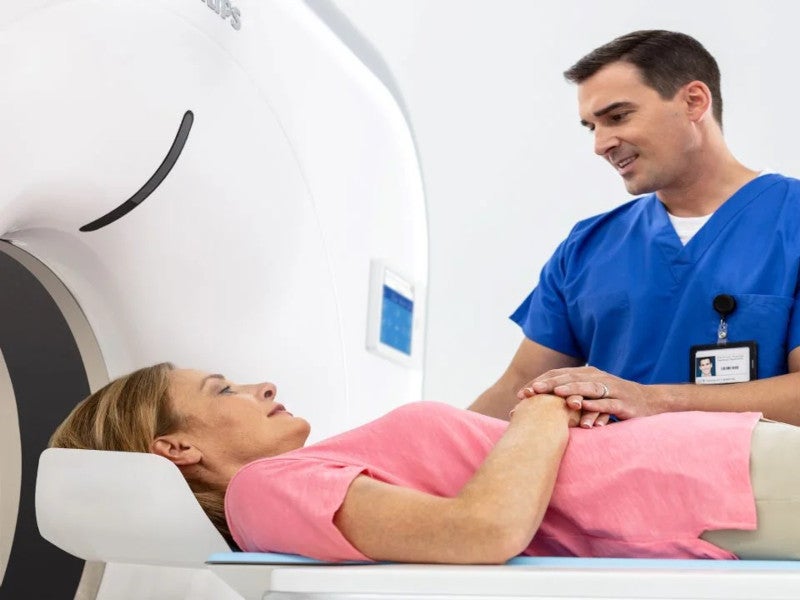
The Incisive CT scanner is a computed tomography (CT) system developed by Netherlands-based health technology solutions provider Royal Philips.
The device reconstructs X-ray transmission data captured at different angles and planes to produce images of the head and body. The scanner helps diagnostic imaging healthcare organisations and imaging departments meet their financial, clinical and operational objectives.
Philips unveiled the Incisive CT platform at the 2019 European Congress of Radiology in February 2019. The device won the Red Dot Design Award, awarded by Germany-based Red Dot, later that year. The scanner was installed at Anson Bay Medical Centre in Malaysia in January 2021.
In March 2021, the company launched its artificial intelligence (AI)-enabled Precise Suite solution for the Incisive CT scanner. The solution enhances CT workflows to improve diagnostic confidence, as well as the experience of patients and staff.
Incisive CT has received the CE mark of approval and obtained 510(k) clearance from the US Food and Drug Administration (FDA).
In November 2023, Philips launched mobile CT lung screening trucks to improve the detection and treatment of lung cancer and other respiratory ailments in the US and Australia.
Equipped with the Incisive CT scanner, the mobile units aim to provide lung screening services to high-risk patients and people in remote and rural areas.
Incisive CT scanner design
The Incisive CT scanner has a bore size of 72cm and a detector array with a 50cm scan field of view.
It is available in two configurations, one with a 4cm-wide detector and another with a 2cm-wide detector. It comes with 32, 64 and 128-slice configurations.
The scanner incorporates advanced technologies in imaging, workflow and life-cycle management. The system can be combined with PerformanceBridge, an integrated portfolio of services and solutions to help organisations achieve continuous process improvement.
The scanner uses a proactive monitoring system to provide 24/7 monitoring for predicting and solving issues before they impact day-to-day operations. It also uses Philips’ Remote Services technology to fix 31% of all issues without the need for on-site service.
Features of the Incisive CT scanner
Incisive CT features DoseWise Portal, a web-based dose monitoring solution that collects, measures, analyses and reports radiation exposure of patients and staff to help control the quality of care, efficiency and safety of patients and staff.
The scanner also features the company’s iDose4 Premium Package, which can improve image quality.
The package comprises iDose4, a technology for artefact prevention and increased spatial resolution at a low dose, and O-MAR, a technology for reducing artefacts caused by large orthopaedic implants. iDose4 and O-MAR work together to produce high image quality with reduced artefacts.
When operated in the 70kV scan mode, the scanner improves low-contrast detectability at low doses without sacrificing image quality, thus providing clinical confidence among staff for managing doses.
Incisive CT includes OnPlan, a patient-side touchscreen gantry controls feature. This reduces the time spent away from the patient by enabling the radiologist to carry out several tasks, including setup and pre-scan adjustments, from the patient’s bedside.
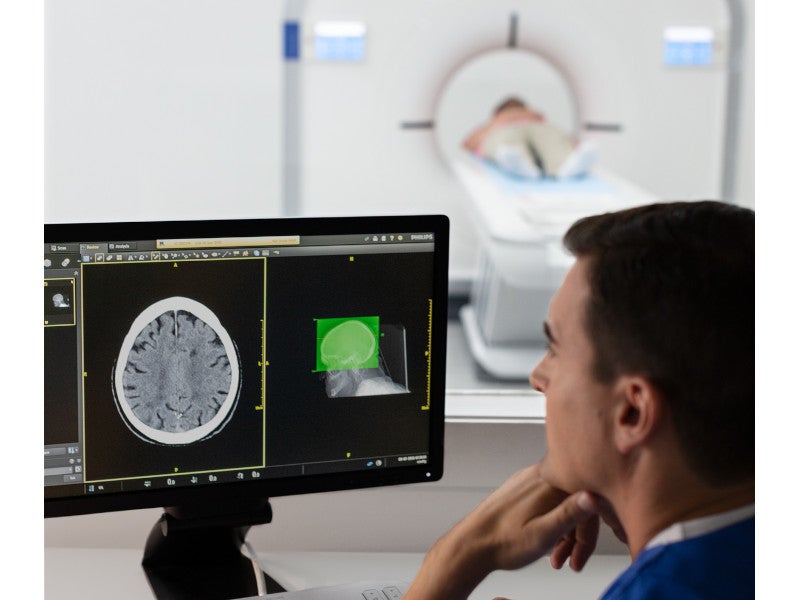
The Incisive CT scanner can be integrated with Philips’ IntelliSpace Portal for post-processing complex cases and giving radiologists access to advanced visualisation capabilities.
vMRC tube details
The Incisive CT scanner features Philips’ vMRC X-ray tube, which is made and tested at the company’s Innovation and Manufacturing Centre of Excellence in Hamburg, Germany. The tube’s spiral groove bearing provides focal spot stability and enables continuous tube cooling. Its liquid-metal bearing design is virtually wear-free while the slotted anode works with the spiral groove bearing to contribute to increased tube life and reliability.
The vMRC Smart Card captures critical performance metrics and operational data and connects to remote 24/7 monitoring and maintenance.
The Incisive CT is being offered with a ‘Tube for Life’ guarantee, in which Philips will replace the X-ray tube throughout the system’s entire lifespan at no added cost. This could potentially reduce operating costs by an estimated $420,000.
To reduce the cost of obsolescence, the system will be available with Philips’ Technology Maximiser programme, which lets users receive the latest software and hardware updates as they are released.
AI-enabled Precise Suite details
The AI-enabled Precise Suite delivers smart workflows from image acquisition through reporting with AI-enabled image reconstruction. It provides motion-free cardiac image capture, automated patient positioning, and real-time interventional guidance to drive precision in dose, speed, and image quality.
Precise Suite addresses the challenges of increasing patient volumes and the need for consistent image quality using AI to streamline and automate workflows and maximise image quality.
It reduces the number of re-scans required and improves diagnostic confidence.
Benefits of Incisive CT
A study performed at Oz Radiology Group revealed that the system reduced scanning time by 19%. The system enables inter-operator consistency and accommodates a range of patient types and exams. The adaptive intelligence allows it to produce high-quality and fast results consistently.
The high-quality images produced by the scanner enable radiologists to make more accurate diagnoses, leading to better patient outcomes.
The advanced imaging techniques used in the Philips Incisive CT Scanner allow for reduced radiation doses, cutting the risk of radiation exposure for patients.