The Eversense® continuous glucose monitoring (CGM) system is a device used to monitor blood glucose levels in people with diabetes.
The device was developed by Senseonics and is one of the first approved long-term CGM systems. It can monitor blood glucose levels every five minutes for up to 90 days.
Eversense was approved by the US Food and Drug Administration (FDA) as a prescription device in June 2018. The device was commercially launched in August 2018.
Horizon Blue Cross Blue Shield and Horizon Healthcare of New Jersey became the first commercial provider of the device under its medical policy.
Eversense components and usage
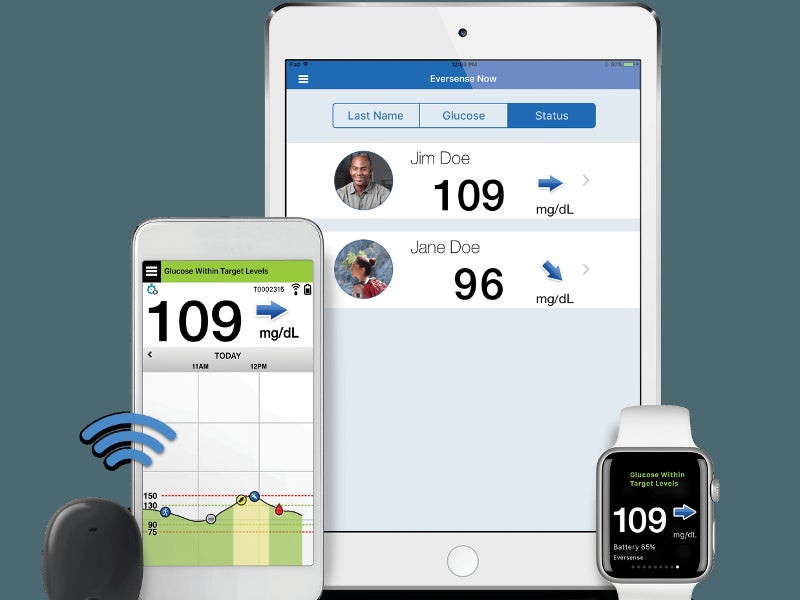
The Eversense CGM system provides blood glucose level readings from 40mg/dl to 400mg/dl in real-time. The system consists of a subcutaneously implantable sensor, a removable transmitter and a mobile app.
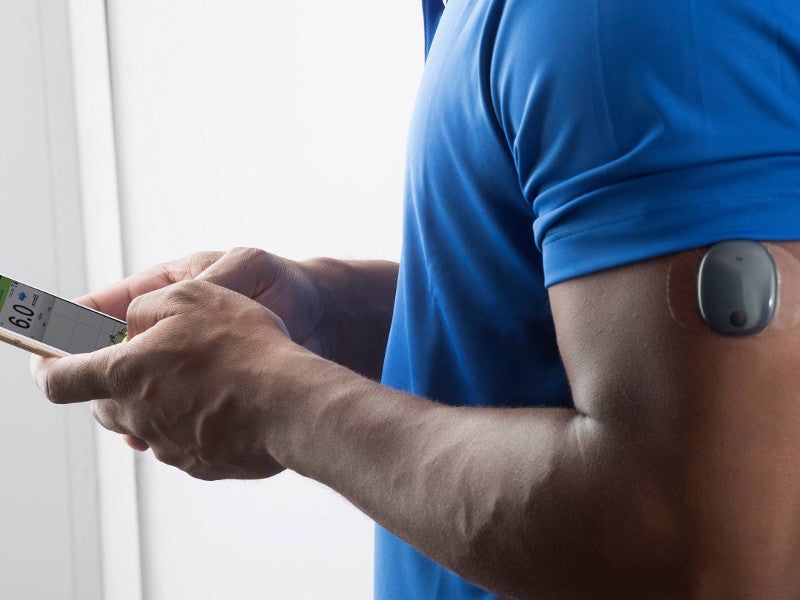
A single implantable sensor coated with a fluorescent polymer is implanted subcutaneously in the upper arm of the patient. It responds to the glucose concentration present in the interstitial fluid of the body and emits a small amount of fluorescent light.
A transmitter is worn over the sensor on the body, which receives the blood glucose level reading from the sensor and calculates the value of blood glucose. The blood glucose value is then transmitted to the mobile app via Bluetooth.
The mobile app sends alerts to the user about the fluctuating glucose values sent by the sensor. It also tracks daily activities such as intake of meals and exercise to provide a graph for identifying trends and patterns.
Eversense CGM system sensor details
The implantable sensor is made of biocompatible materials, including homopolymer polymethylmethacrylate, hydroxyethylmethacrylate-based hydrogel, platinum, silicone, and epoxy 301-2. It measures 18.3mm in length and 3.5mm in diameter and is sterilised by ethylene oxide.
The sensor features a silicone elution ring containing a small amount of dexamethasone acetate, an anti-inflammatory drug. When the sensor comes into contact with the interstitial fluids, the ring releases a small amount of the drug, which helps reduce the body’s inflammatory response against the implanted sensor.
Details of the smart transmitter in the Eversense system
The transmitter in Eversense continuous glucose monitoring system is a round-shaped, water-resistant device with a polycarbonate body. It is designed to provide on-body vibratory alerts when the blood glucose levels fluctuate, even when the mobile device is away.
The transmitter also powers the sensor and contains a non-replaceable, rechargeable lithium-polymer battery, which can be reused for up to a year.
The transmitter is 37.6mm long, 48mm wide and 8.8mm thick. It offers a communication distance of up to 24.9ft between the app and the transmitter.
Benefits of CGM technology
The global prevalence of diabetes has increased from 108 million in 1980 to 422 million in 2014. According to the World Health Organisation (WHO), there were around 1.6 million deaths directly related to diabetes worldwide in 2016.
CGM systems enable efficient diabetes management and blood glucose control in people, thus improving the quality of life. The conventional blood glucose meter provides blood glucose level readings only for a particular moment, while the Eversense CGM system offers readings in real-time.
The Eversense CGM system also furnishes glucose trend information and alerts for detecting and predicting low or high blood glucose episodes. Blood glucose data is stored in both the mobile app and the transmitter.