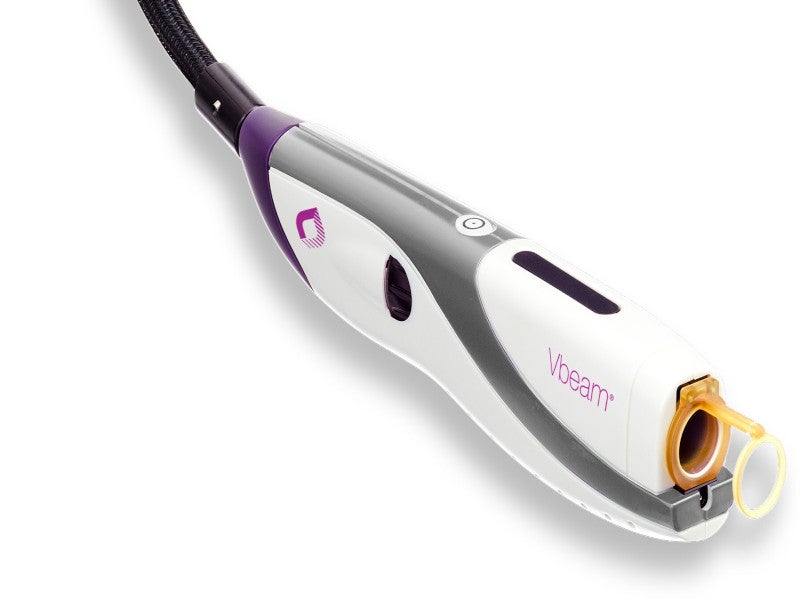
The Vbeam® Prima™ is an advanced 595nm pulsed-dye laser (PDL) system developed by Candela, an aesthetic device company based in the US, to treat vascular and dermatologic conditions.
The device is used to treat a wide range of skin conditions such as acne, benign cutaneous lesions, cutaneous capillary malformations, infantile haemangiomas (IH), pigmented and non-pigmented lesions, rosacea, scars, stretch marks, wrinkles and warts in infants and adults.
Regulatory approvals for Vbeam Prima PDL
The US Food and Drug Administration (FDA) approved the Vbeam Prima 595nm pulse-dyed lasers system (PDL) in July 2018. The device further became commercially available in Canada in April 2019.
In June 2023, the Vbeam family of PDL received FDA clearance to include paediatric patients from birth to 21 years of age to treat cutaneous capillary malformations, also called port wine stains (PWS), and IH. The device also holds the CE mark in Europe.
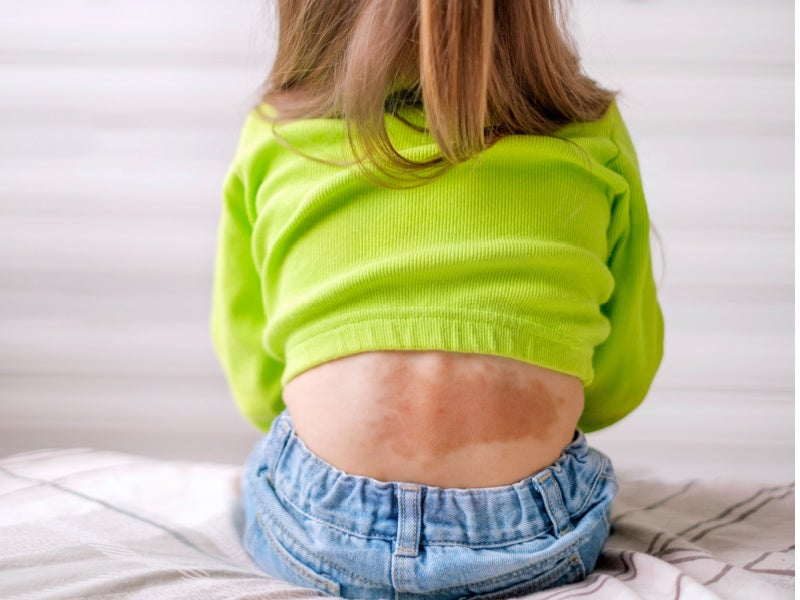
Cutaneous capillary malformations and infantile haemangiomas disorder
A flat reddish-pink birthmark on a child’s skin is called a capillary malformation, which is a vascular malformation caused by enlarged capillaries. It increases and darkens as children get older, and produces vascular abnormalities.
One in 330 babies is born with a capillary malformation, and early treatment is key for the best results.
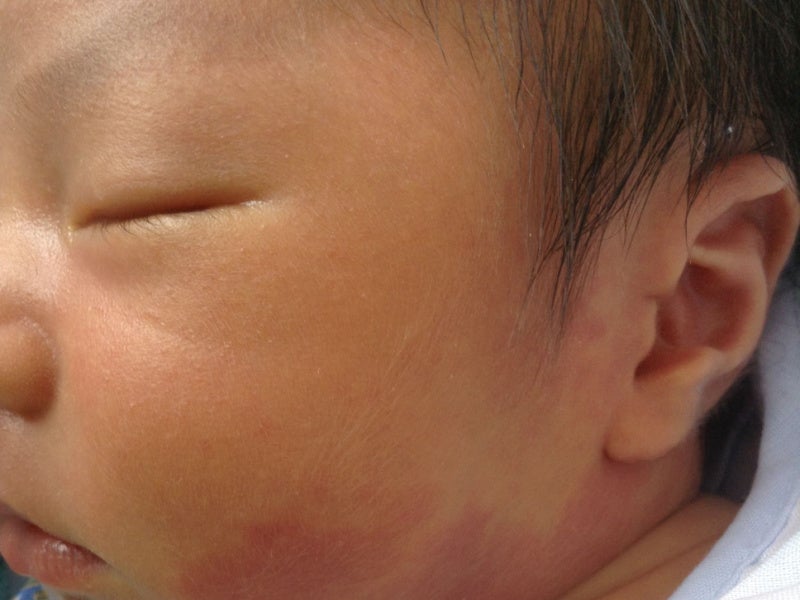
IHs are blood vessels that form improperly and multiply excessively, causing rapid growth of blood vessels in babies. The condition develops mostly after delivery or birth.
The most effective approach for treating haemangiomas is the Vbeam 595nm PDL, which has no negative side effects and can stop growth and improve regression.
Vbeam Prima design and features
The Vbeam Prima features the proprietary 595nm wavelength and an additional 1064nm wavelength with new additions including two dermal cooling options.
The 595nm wavelength penetrates the skin to target specific blood vessels. Oxyhaemoglobin absorbs its energy to coagulate and clear vessels with more tolerability and lesser melanin absorption. The 1064nm wavelength targets deep blue veins, venous lakes and wrinkles.
The Vbeam Prima system features a large treatment spot size of 15mm and administers 12J [joule] of radiation at the 595nm wavelength, ensuring fast and effective treatment.
Two cooling options offer maximum adaptability and epidermal protection. The cryogen-based Dynamic Cooling Device™ (DCD™) adapts fluidly to automatically provide reliable epidermal protection. The vascular and pigmented lesions are treated with EverCool™ contact cooling.
The Vbeam Prima remains patient and treatment-ready throughout the day, minimising patient waiting times and reducing calibration to first use per wavelength.
The device’s dye life meter offers real-time data to help users avoid unanticipated treatment interruptions attributed to dye loss. The adaptive zoom handpiece of the device allows precise targeting by adjusting the spot size in increments as small as 0.5mm.
Wi-Fi connectivity provides for quicker equipment service and remote diagnostics, while user-friendly software makes it easy to access previously recorded treatment settings.
Vbeam Prima 595nm applications
The Vbeam Prima 595nm is used for photocoagulation of benign cutaneous vascular lesions including angioma, facial and leg telangiectasia, haemangiomas, poikiloderma of civatte, PWS, rosacea and spider angioma, as well as benign cutaneous lesions such as warts, scars, striae, psoriasis and wrinkles. It also treats benign epidermal pigmented lesions and inflammatory acne vulgaris.
It is used in general surgery, dermatology or plastic surgery, gynaecology and podiatry.
Vbeam Prima 1064nm applications
The Vbeam Prima 1064nm treats benign vascular lesions including haemangiomas, leg veins, poikiloderma of civatte, PWS, rosacea, spider veins, telangiectasia venous lakes and warts.
It also treats benign cutaneous lesions such as cafe au lait macules, chloasma, keratoses, lentigos (age spots), nevi, periocular and perioral wrinkles, seborrheic keratoses, skin tags, solar lentigos and verrucae.
The treatment helps in the coagulation and haemostasis of these lesions.
Clinical benefits of Vbeam Prima
The Vbeam Prime system offers several benefits, including visibly reducing rosacea symptoms from moderate to mild and reducing difficult-to-treat erythema from severe to mild.
Approximately 84% of patients experienced over 40% improvement in rosacea, with minimal adverse side effects, in a clinical study. The Vbeam® Prima system, with a 595nm wavelength, targets the vascular component of scar tissue, improving cosmesis and reducing pain.
In a clinical study, 100% of scars subjected to the Vbeam 595nm PDL exhibited improvements in cosmesis. Additionally, 80% of patients undergoing Vbeam PDL treatment reported high levels of satisfaction, ranging from very to extremely satisfied, while 77% reported experiencing little to moderate pain.
The Vbeam 595nm PDL is proven effective for treating vascular skin lesions and improving the cosmetic appearance of surgical scars in Fitzpatrick skin types I to IV.
It has been found to be both safe to be used on more than 6,000 paediatric patients with haemangiomas and strong enough to provide long-lasting effects in trials carried out globally.