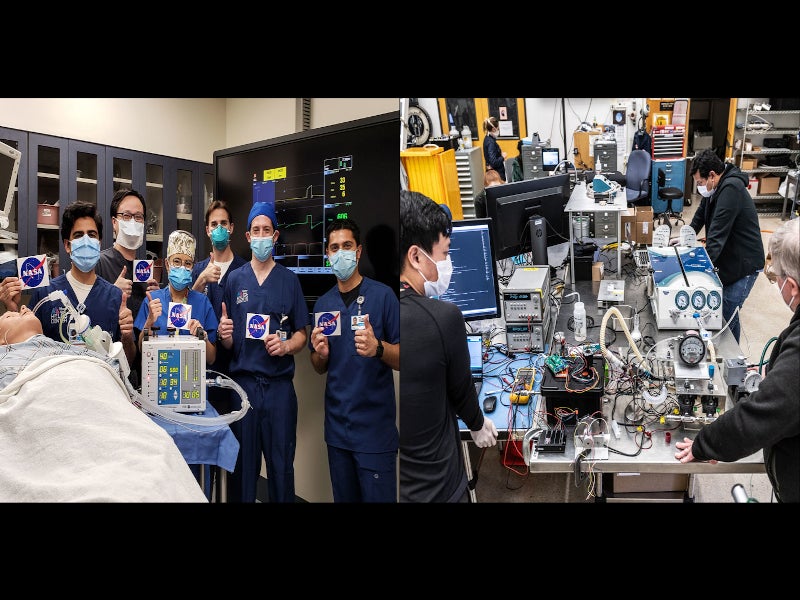
The Ventilator Intervention Technology Accessible Locally (VITAL) is a new high-pressure ventilator developed by the National Aeronautics and Space Administration’s (NASA) Jet Propulsion Laboratory (JPL) in Southern California to treat patients with severe Covid-19 symptoms.
JPL is managed by the Office of Technology Transfer and Corporate Partnerships (OTTCP) at the California Institute of Technology (Caltech).
The device was engineered to support respiration in Covid-19 patients with acute respiratory distress syndrome (ARDS). The prototype was developed in just 37 days and successfully tested, using high fidelity human patient simulator on 21 April 2020 at the Icahn School of Medicine at Mount Sinai in New York.
An emergency use authorisation (EUA) application was submitted to the US Food and Drug Administration (FDA) for the device on 22 April 2020.
VITAL secured FDA’s ventilator EUA on 30 April 2020. It was placed in the list of Authorized Ventilators, Ventilator Tubing Connectors, and Ventilator Accessories, issued under the ventilator EUA.
VITAL device details and features
VITAL is a simple, mass manufacturable ventilating device, which functions as traditional ventilator but contains fewer components when compared to a conventional ventilator.
It is a control device that assists in providing volume targeted, pressure set, and time-limited ventilation for a broad range of parameters set by the healthcare professionals.
With the supplemental oxygen capability between 21% and 100%, the positive end-expiratory pressure (PEEP) value of the device is 5cm to 35cm H2O with the back-up rate of four to 40 breaths per minute and tidal volume between 150ml and 800ml.
Patients who require respiratory support are sedated and an oxygen tube is inserted into their respiratory passage for breathing.
VITAL ventilation device can run continuously for 20 days without maintenance. It has the operational lifetime of four months with multiple patient utilisation before disposal. VITAL can be stored for at least 24 months in a controlled environment.
The device can be moved within a facility by connecting it to an uninterruptible power supply (UPS) for operation during transit and can withstand dirt and dust of the hospital setting, operating in a specific environment with relatively considerable temperature and humidity exposure.
VITAL device technology
The device is based on a simplified, low-cost ventilator-system technology. NASA’s JPL developed two prototypes of the VITAL, a pneumatic ventilator and a compressor ventilator targeted to treat symptomatic Covid-19 patients, requiring pre-ICU respiratory support in different clinical states including high-resistance, low-compliance, hypoxia and hypercapnia.
Licensing details of VITAL
Office of Technology Transfer and Corporate Partnerships offers free non-exclusive technology license to industry production partners for rapid manufacturing, commercialisation and import of the device to meet the domestic and global demand during the Covid-19 pandemic or till 1 October 2024, whichever is earlier.
The license will include detailed technical information package and limited technical assistance.
Suitable applicants will receive the license and the US government intends to purchase tens of thousands of the device in 2020.
Advantages of VITAL device
The device is fitted with minimal mechanical and electronic components to reduce the assembly time and increase the cost-efficiency. Patient exhalation gases are removed from the circuit, avoiding the device sterilisation process before using it for the next patient.
VITAL flexible design can be modified according to the operational requirements of the setting, such as hospitals, to be set up in hotels or other places. The device will not pose a threat to the existing hospital ventilators as the latter last for several years and can address a range of medical conditions.
The highly dynamic VITAL ventilation device will easily support the advanced stage Covid-19 patients in ICU, who require advanced ventilator support.