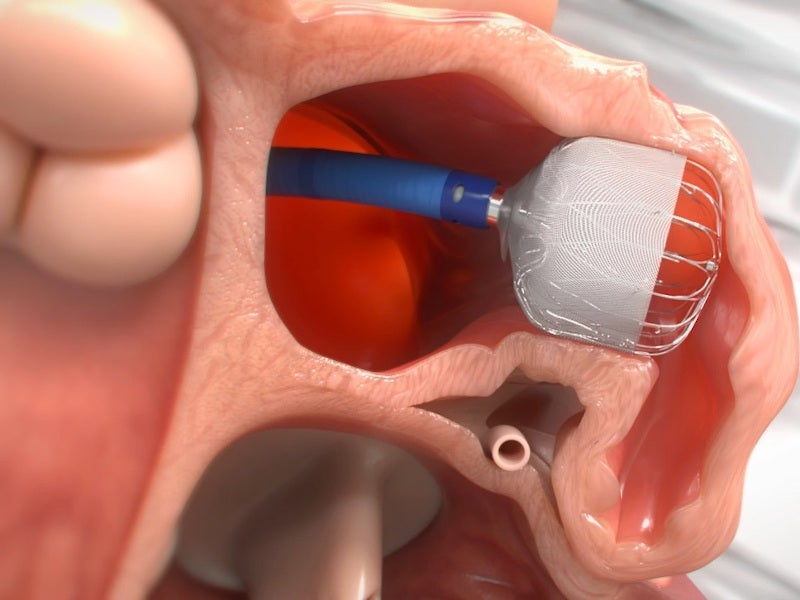
The WATCHMAN FLX™ is the first FDA-approved left atrial appendage closure (LAAC) device developed by Boston Scientific, a medical device company based in the US.
The device is indicated to minimise the risk of stroke in non-valvular atrial fibrillation (NVAF) patients who require an alternative to oral anticoagulation therapy by permanent closure of the left atrial appendage (LAA). The LAA is a region in the heart where stroke-causing blood clots are typically generated in NVAF patients.
Boston Scientific received the European CE Mark for the WATCHMAN FLX LAAC device in November 2015 while its next-generation device received the CE Mark in March 2019. The FDA approved the device in July 2020.
In September 2022, the FDA approved expanded labelling for the device. The expansion included a 45-day dual antiplatelet therapy (DAPT) option, providing an alternative to the 45-day oral anticoagulation (OAC) plus aspirin for post-procedural treatment in patients with NVAF.
Boston Scientific received approval for the latest version of the device, WATCHMAN FLX Pro, from the FDA in September 2023. The new device is coated and designed to resist thrombosis and is equipped with new features to enhance procedural performance and safety. It has been successfully used in approximately 190,000 of more than 300,000 WATCHMAN procedures worldwide.
WATCHMAN FLX LAAC device details
The WATCHMAN FLX is a next-generation LAAC device with a fully rounded frame design for implantation in patients in a one-time procedure. It is the first LAAC device that can be completely grasped, rearranged and redeployed for accurate positioning.
The design of the new frame facilitates superior device engagement for long-term stability and ensures a rapid and comprehensive seal. It allows the physicians to reach the device easily and safely within the LAA for manoeuvring during the procedure.
The device employs new stroke risk-reducing technology to improve its performance and safety during the procedure, as well as treat a range of patients with NVAF.
The WATCHMAN FLX LAAC system acts as a one-time solution to blood thinners in NVAF patients to minimise stroke risk.
The device is available in multiple size options than the previous generation device to suit varying anatomies.
WATCHMAN FLX device functionality
Atrial fibrillation (AF) affects the heart’s capacity to pump blood naturally, which can lead to the accumulation of blood in the LAA of the heart.
The blood accumulation in LAA increases the risk of blood clot formation in the area, which can gradually migrate to other parts of the body and cut off the blood supply to the brain, causing a stroke.
More than 90% of stroke-causing clots in the heart are formed in the LAA of NVAF patients and closure of the LAA is a successful procedure to minimise the risk of stroke in affected patients.
The WATCHMAN FLX device is implanted via a transfemoral approach, using a minimally invasive procedure. It is designed to completely close the LAA, which can allow many patients to avoid long-term use of blood thinners while minimising their associated bleeding risk and lifestyle challenges.
WATCHMAN FLX Pro features
The new WATCHMAN FLX Pro device is built on the established safety and procedural performance of the WATCHMAN FLX device. It is used for percutaneous transcatheter closure of the LAA.
The device incorporates HEMOCOAT™ technology, which is a durable, thromboresistant polymer coating that reduces inflammation and promotes quicker and more thorough endothelialisation. The coating also minimises device-related thrombus formation and facilitates quicker and more controlled healing.
The new device incorporates three new radiopaque visualisation markers to improve device placement for optimal sealing around the LAA. It is available in a larger 40mm size with a broader size matrix to treat a wide range of anatomies.
The WATCHMAN FLX Pro device mitigates the risk of thromboembolism from the LAA in patients with NVAF prone to increased risk for stroke and coagulation based on CHA2 DS2-VASc1 scores and is recommended for anticoagulation therapy.
Clinical study on WATCHMAN FLX device
The PINNACLE FLX pivotal trial determined the procedural safety and closure efficacy of WATCHMAN FLX LAAC system.
The single-arm, non-randomised trial evaluated the device as a substitute for oral anticoagulation therapy, including non-vitamin K antagonist oral anticoagulants (NOACs), for stroke risk reduction in patients with NVAF. It was carried out on 400 patients at 29 locations across the US.
The rate of successful LAA closure was the primary endpoint of the trial, which was defined as any peri-device flow less than or equal to 5mm at 12 months shown by the transoesophageal echo (TEE) test.
The WATCHMAN FLX device achieved a low rate (0.5%) of adverse events seven days after the procedure and a higher rate (100%) of effective LAA closure at 12 months. The data also showed a high (98.8%) implant success rate for the device.
Clinical study on WATCHMAN FLX Pro device
The WATCHMAN FLX Pro demonstrated positive results in multiple preclinical research for faster, controlled healing around the device surface.
The data from the preclinical studies showed that the polymer coating resulted in an 86% decrease in inflammation three days following the procedure, a 70% thrombus reduction at 14 days and a 50% increase in endothelial tissue coverage at 45 days after the procedure.
The WATCHMAN FLX Pro is currently being evaluated in the WATCHMAN FLX Pro CT study, a premarket study conducted at a single centre. The study utilises various imaging modalities to evaluate the coverage of device tissue after the procedure and its potential connection to clinical outcomes.
Additionally, the device will undergo further assessment in the post-market HEAL-LAA study, which will monitor outcomes in around 1,000 patients with NVAF who have been implanted with the technology at 60 sites across the US.