Technological advancements in the US healthcare sector are increasing all the time. With demand growing and technology getting smarter, the effects are being felt throughout the medical device industry. The presence of major medical devices companies is anticipated to boost fine medical wire demand in the country, with the fine medical wire market set to expand exponentially in the next ten years.
According to a global market study by Persistence Market Research, the fine medical wire market is predicted to expand at a healthy CAGR (compound annual growth rate) of more than 6% over the 2020-2030 assessment period.
Cardiovascular disorders place a huge strain on the healthcare system and are increasing at an alarming rate. According to a research report by Global Banking and Finance Review on the fine medical wire market: “Rising prevalence of CVD is driving demand for fine medical wire for guide wires, catheter reinforcements, and pacing leads, which act as integral parts of medical devices.”
Custom Wire Technologies (CWT) is a leading OEM medical component manufacturer, with over 50 years of industry experience and implementation of the latest technologies.
CWT manufactures custom fine wire, ultra-fine wire, and guidewire medical components for various devices and components using a wide range of wire materials. The company specialises in the production of prototypes as well as contract production orders for OEMs, medical device manufacturers, and researchers.
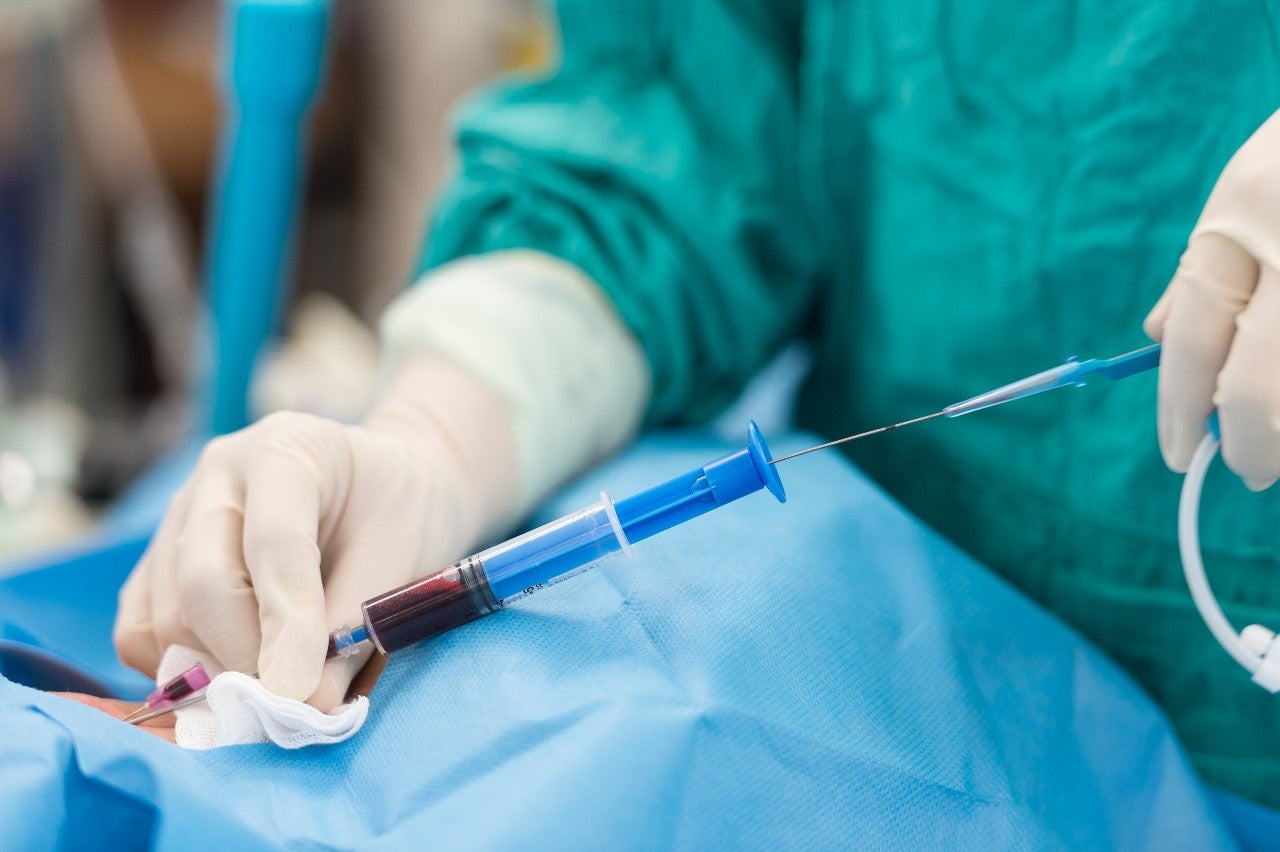
Change in treatment
In recent years there has been a large shift to minimally invasive or non-invasive surgical treatments, particularly in geriatric patients. Minimally invasive procedures reduce the size of incisions required in surgery, thereby reducing the healing time of wounds and associated risks of infection.
Through this method of surgery gaining more support, the demand of guidewire for treatments is significantly increasing, encouraging more companies to invest and develop in advanced guidewires for a variety of medical applications.
Fine medical wire is also being more frequently used in less invasive orthopaedic surgeries. Persistence Market Research claims this trend is likely to continue and will be further enhanced by innovative technologies, making surgery more precise and orthopaedic surgeons an increasingly proactive specialist.
For CWT, all this has materialised in an increased demand for its smaller fine wire products – coiled and ground. At the same time, as the need for improved treatments strengthens, the component supplier is facilitating some much-needed innovations in the medical devices industry.
“Today physicians are saying ‘if I was able to do this, I could help to treat this patient’. Many times, the tools to help in the treatment are not available or have not been developed. This is where CWT and our capabilities come into play. Our goal is to assist in coming up with solutions. We are motivated in making a difference and saving lives,” says Bob Boldig, president of Custom Wire Technologies.
COVID-19 impact and the future of the industry
The outbreak of coronavirus initially impacted the revenue of fine medical wire manufacturers due to the disruption in demand which jolted market growth. With more surgeries happening daily but hospitals wanting to keep recovery time and readmittance for pain and infection to a minimum, the fine medical wire market is facing an increased demand in production.
CWT is taking measures to ensure it’s ready to meet this growing demand. “We have doubled our physical size to allow for continued processing equipment expansion,” says Boldig. “We are also strategically working with our raw material suppliers to control the long lead times that have developed to give us the ability to produce components quickly.”
Aside from that, the company plans to stay close to its core competencies in coiling and grinding, where it sees strong, sustained growth. In particular, nitinol coiling, shape setting, and grinding continue to grow at a rapid pace and have all become niches for CWT.


