When discussing medical devices such as pacemakers and implantable cardioverter defibrillators (ICD), often people who have been unaffected assume that these devices are only necessary for the elderly. What’s more, they may assume that living a healthy lifestyle ensures that you will not need one.
Danish footballer Christian Eriksen suffered a cardiac arrest during Euro 2020. As worried family, friends and fans looked on, he was resuscitated on the pitch. Fortunately, he recovered, and was promptly fitted with an ICD – a small device that can treat people with dangerous abnormal heart rhythms by sending electrical pulses to the heart.
As a 29-year old professional athlete, many were shocked at his sudden collapse, but this isn’t the first time that something like this has happened. In 2012, English footballer Fabrice Ndala Muamba suffered cardiac arrest and collapsed during the first half of an FA Cup quarter-final match. Cameras kept a respectable distance as Muamba was resuscitated on the pitch with numerous defibrillator shocks. He was later fitted with an ICD. After Eriksen’s collapse, Muamba expressed his sympathies and support in a BBC radio interview.
In 2008, Beligian footballer Anthony Van Loo was fitted with an ICD following the diagnosis of a heart condition. Fans and teammates were shocked when he suffered cardiac arrest during a match the following year, but onlookers saw how the ICD device saved his life. Video footage shows Van Loo’s leg twitching after he collapsed, which indicated that the ICD was firing. Without this medical device, immediate medical intervention is required – as was the case with Muamba and Erikson.
Thanks to the hard work of field-side medical teams and hospital staff, these three men have recovered and are living their life with their ICD, but how do these devices work?
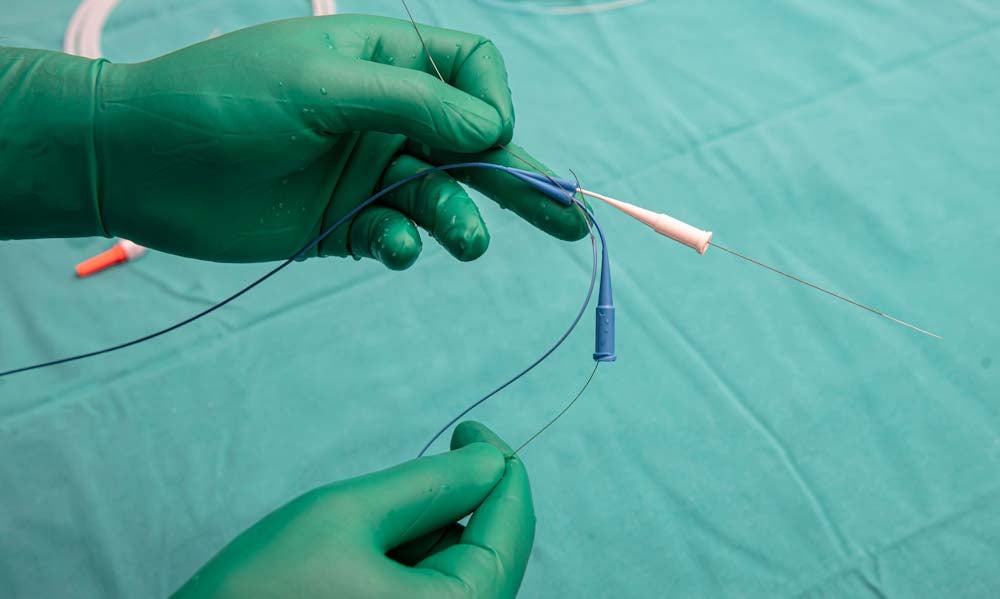
An ICD is placed under the skin, typically in the space below the clavicle, in order to monitor heart rate. Thin medical wires connect the ICD to the heart, where it continually checks heart rate and rhythm using electrodes. If the ICD picks up a dangerous heart rhythm, it can intervene in several ways. For pacing, it delivers a series of fast-paced, low-voltage electrical impulses to correct the heart rhythm. For cardioversion, it delivers one or more small electric shocks. For defibrillation, the treatment that saved these young footballers’ lives, one or more larger electric shocks are delivered to restore a healthy heart rhythm.
Implantable cardioverter defibrillators are used by a wide range of people who have experienced a life-threatening abnormal heart rhythm and are at risk of it happening again, or whose tests have shown they are at risk of one in the future, often due to inherited conditions such as Cardiomyopathy, Long QT syndrome or Brugada syndrome.
Patients who have heart failure and are at risk of developing a life-threatening heart rhythm require cardiac resynchronisation therapy with a defibrillator (CRT-D), which is a device that combines a pacemaker with an ICD.
Companies such as Sandvik manufacture medical device wire used in ICDs and pacemakers, by combining metallurgy, process development methods and coatings to configure components that are perfectly calibrated for the application.
According to Lena Wiig Boström, Marketing Communications Manager at business unit Medical: “It’s very rewarding for us at Sandvik to know that the wires we make are used in applications such as heart starters or heart pacemakers. They both save lives and make it possible for people to live a normal life”.
For more information about EXERA® fine medical wire, including a full list of materials or to contact Sandvik about developing a medical field device, visit their website or download the whitepaper below.