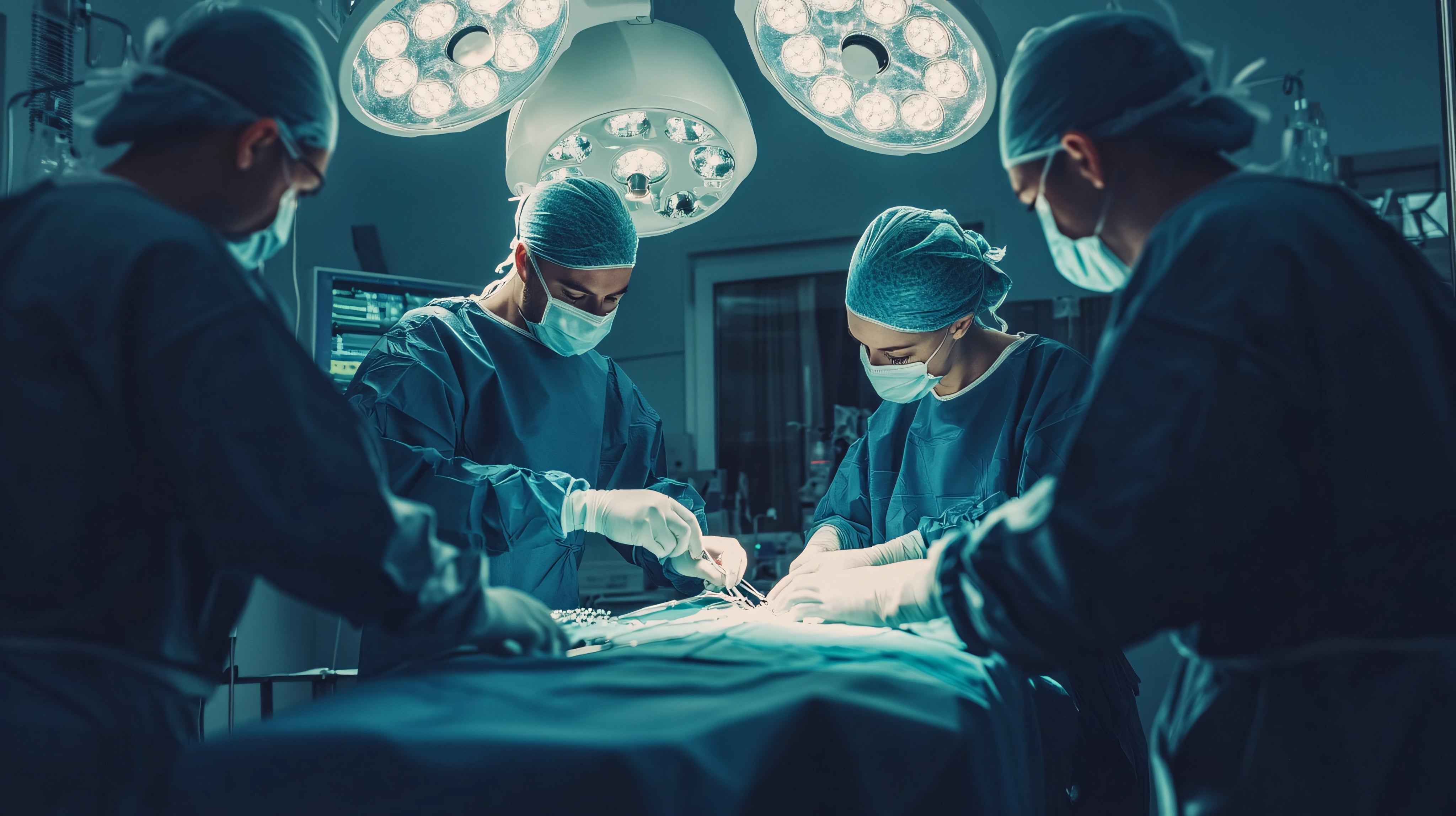
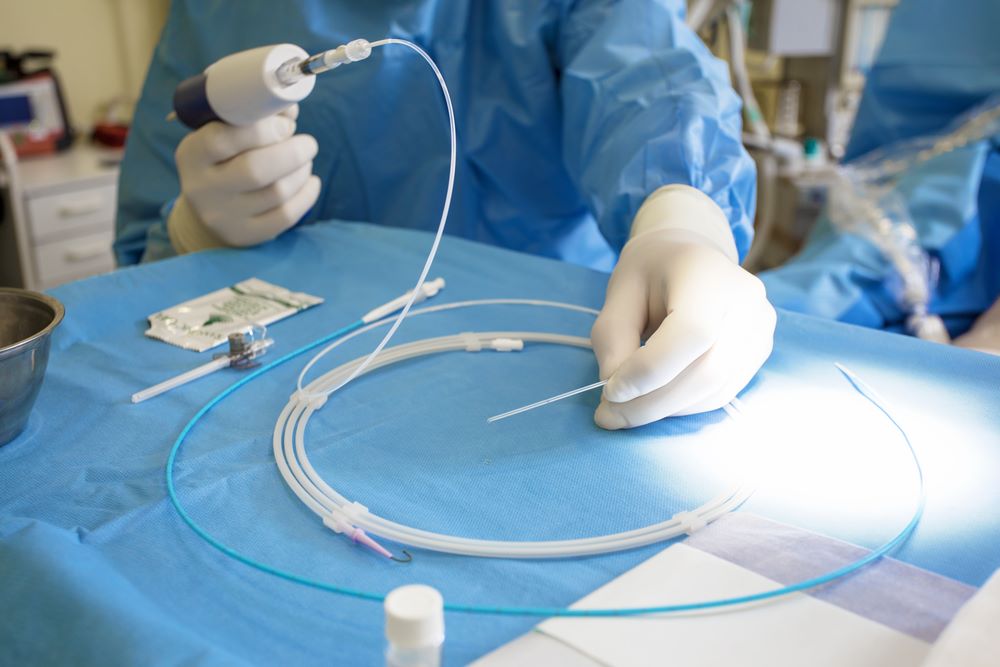
Offered as an alternative to drug-based treatments, catheter ablation is used to treat abnormal heart rhythms (arrhythmias), especially when the condition’s symptoms are interfering with the patient’s quality of life.
The process involves passing catheters through the blood vessels to the heart where it records the heart’s electrical activity and ascertains where the arrhythmia is coming from. This affected area of the heart is then damaged using heat (radiofrequency ablation) or freezing (cryoablation) to create scar tissue. As scar tissue does not conduct electricity, the problem area cannot receive electrical signals that reach the rest of the heart.
This procedure can be used for supraventricular tachycardia (SVT) treatment, which is a condition where the electrical system that controls your heart rhythm is not working correctly, causing resting heart rates well above average.
There are many stringent requirements when manufacturing medical wire for ablation leads used for SVT treatment. As it is passing through the body to the heart, it must be very flexible and responsive to turning in order to make very precise movement and not cause any damage. Additionally, it must be lubricious and constructed from a material that does not flake off or become a contaminant.
Meeting all of these requirements can be tricky, so many OEMs enlist the help of experts such as Sandvik to design and develop medical wire components. Under its EXERA® brand, Sandvik develops alloys and stainless-steel wire with fatigue relaxation resistance, 1:1 torque for precise movement, moderate tensile strength and anti-corrosion properties, as well as PTFE Teflon coatings that give wire non-stick, low-friction properties without the risk of pollutants. For catheters, Sandvik also manufacturers wire in the form of braid-reinforced shafts.
For more information about EXERA® fine medical wire-components, including available materials and surface treatments, download the white paper below.