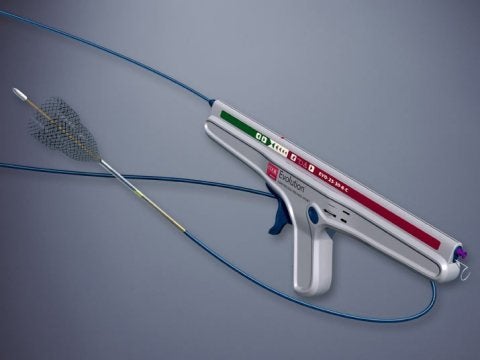
Cook Medical has received US Food and Drug Administration (FDA) approval for its new Evolution colonic controlled-release stent, designed to relieve large-bowel obstructions prior to colectomy procedures.
The new stent is used to reduce pain and uncomfortable symptoms associated with colonic obstructions including abdominal pain, vomiting, nausea, difficult defecation and GI tract bleeding.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The Evolution stent features a delivery system utilising kink-resistant Flexor technology for pushability and stability, as well as a visible endoscopic marker to define the proximal end of the stent.
The approval was based on a recent international, multicentre study of 80 patients, which was designed to demonstrate the safety and efficacy of the Evolution stent in reducing symptoms, as well as acting as a bridge to surgery.
The first post-procedure report revealed that 85% of patients in both groups had mild or no symptoms of abdominal pain, no difficulty in defecating or constipation, no adverse events associated with device malfunction, as well as no deaths considered to be caused by a device complication.
Cook Medical endoscopy division global business unit leader, Barry Slowey, said the new Evolution colonic stent reiterates the company’s commitment to develop a full line of products to improve the quality of life in patients who suffer discomfort or pain.
“Now clinicians have the ability to precisely deploy and recapture colonic stents,” Slowey added.
“That can make it easier to place the stent the first time around, potentially reducing the need for repeat procedures, while increasing efficiency in the GI unit.”
The company said that the Evolution colonic stent, which is now available to select physicians across the US, will be widely available in the coming months.
Image: The new Evolution stent is proven to reduce pain and uncomfortable symptoms associated with colonic obstructions. Photo courtesy of: Cook Medical.





