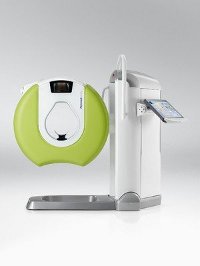
The US Food and Drug Administration (FDA) has granted 510(k) approval to Planmed’s low-dose orthopedic imaging system to provide fast 3D imaging of subtle extremity fractures that have been missed when using 2D radiographs at the point of care.
The CE-marked system, Planmed Verity extremity scanner, uses a significantly lower dose of radiation and adapts to the patient with anatomy-specific imaging programmes, movements and carbon-fibre positioning trays.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The system also includes adjustable user interfaces and an efficient all-in-one workflow to maximise the operator’s soothing presence for the patient.
The scanner, which is intended for pre and postoperative imaging, has a better resolution and patient adaptability compared to full-body computed tomography systems, claims the company.
Planmed Verity’s easily adjustable, soft surfaced gantry and motorised positioning also helps to find a comfortable position for various examination procedures.
Planmed vice president Vesa Mattila said; "Planmed Verity is the first and only CT system in the world that can image seated, supine and standing patients."

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataIn response to the FDA’s clearance of Planmed Verity, the company’s US subsidiary Planmed USA will now sell, market and provide tech support for the product in North America.
Planmed said it will showcase Planmed Verity at the American Association of Orthopedic Surgeons (AAOS) meeting, which will be held in Chicago, the US, from 19 March 2013.
Image: The new Planmed Verity Extremity CT Scanner revolutionises extremity CT imaging and brings low dose 3D imaging to trauma centres and orthopedic clinics for fast diagnosis at the point of care. Photo: Courtesy of PR Newswire.





