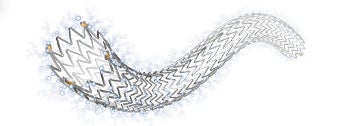
The US Food and Drug Administration (FDA) has approved Cook Medical’s drug-eluting stent for treating peripheral arterial disease (PAD) in the superficial femoral artery.
Initially available in 80mm lengths with 6mm or 7mm diametres, the Zilver PTX drug-eluting peripheral stent includes a paclitaxel drug, which is taken up by the cells of the arterial wall to help prevent the renarrowing of the artery over time.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The stent is also expanded like a scaffold to help keep the artery open after the catheter is withdrawn, according to the company.
FDA approval was based on the randomised controlled study, designed to compare the safety and efficacy of Zilver PTX stent with a bare metal stent.
The study showed that eight out of ten patients treated with the stent still had open arteries (primary patency) after one year.
Stanford University School of Medicine cardiothoracic surgery department professor Michael Dake said: “After conducting the largest randomised controlled study of peripheral stenting ever undertaken, we now see remarkable results in patients treated with Zilver PTX.”
The stent, which is approved for sale in the EU, Japan, Brazil and most of South America, Australia, New Zealand and Taiwan, will also be available in 40mm and 60mm lengths in the US in early 2013.
In addition, the company is expecting to receive approval for 120mm length stents in both diametres within the next year.
Cook Medical peripheral intervention division vice president and global leader Rob Lyles said: “No other company can match Cook’s commitment to this technology, and by the end of 2013, we expect to have a full suite of drug-eluting peripheral stents in the most commonly used lengths and diameters available to US physicians.”
Image: The Zilver PTX drug-eluting peripheral stent including paclitaxel drug treats peripheral arterial disease in the superficial femoral artery. Photo courtesy of Cook Medical.





