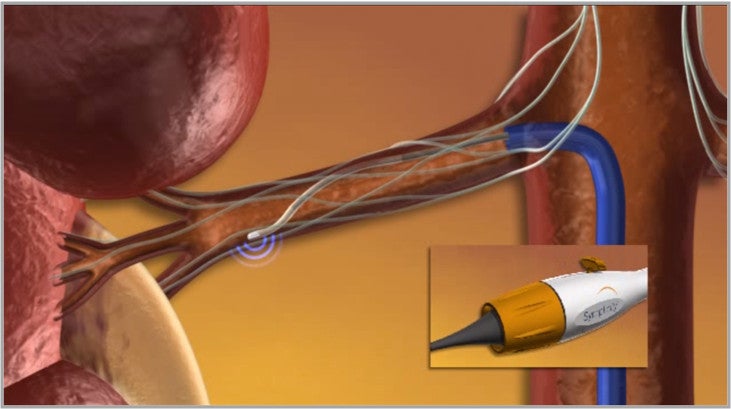
Medtronic has obtained approval in the Europe and Australia to market its next-generation components of Symplicity renal denervation system, designed to reduce ablation time and provide ease of deliverability during renal denervation procedures for patients with uncontrolled hypertension.
The CE Mark and Therapeutic Goods Administration (TGA) approved system includes a highly flexible 4 Fr multi-electrode Symplicity Spyral catheter and Symplicity G3 radio frequency (RF) generator.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The Symplicity Spyral catheter system features an array of four electrodes in a spiral configuration that are able to deliver simultaneous or selective RF energy into the renal artery wall to disrupt the output of overactive sympathetic nerves.
The new 4 Fr catheter is compatible with a 6 Fr guide catheter and is delivered over a 0.014in guide wire via a rapid exchange system.
It is highly conformable to artery shape and size and accommodates vessel diameters of 3mm-8mm.
According to the company, the multi-electrode system is built upon the clinical success and strong safety profile of single-electrode Symplicity renal denervation system.
The Symplicity Spyral catheter is powered by a new RF generator that leverages the benefits of Medtronic’s proprietary Symplicity treatment algorithm with its built-in safety features.
The non-occlusive design ensures the catheter avoids obstruction of renal blood flow during the procedure and allows physicians to turn on and off specific electrodes to accommodate different anatomies.
The G3 generator includes a new touchscreen user interface compatible with the single-electrode Symplicity catheter.
Medtronic Renal Denervation vice-president and general manager Nina Goodheart said: “We have significantly enhanced our technology with sophisticated features designed to meet specific unmet needs and we believe these improvements, coupled with our strong safety and efficacy profile, will provide unprecedented benefit to both patients and physicians.”
The Symplicity renal denervation system is available for investigational use only in the US.
Medtronic’s global Symplicity clinical programme is expected to involve more than 8,000 patients including the US, Europe, and Japan, with more than 1,200 of these patients participating in randomised clinical trials.
Image: Symplicity Renal Denervation System. Photo: courtesy of Medtronic Inc.





