German firm RAUMEDIC has obtained the 510(k) clearance from the US Food and Drug Administration (FDA) to market Neurovent-PTO, a multimodal catheter designed to measure intracranial pressure (ICP), intracranial temperature (ICT) and oxygen partial pressure.
Neurovent-PTO uses the process of ‘oxygen quenching’ and is reportedly the only marketed device that combines three parameters in one catheter.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
By using the oxygen quenching process for the measurement of oxygen partial pressure, the level and changes in the parameters can be measured safely, quickly and accurately.
Oxygen partial pressure measurement records the available oxygen in the brain tissue.
This ensures that any potential cerebral damage is quickly detected and appropriate measures for the avoidance of cerebral ischaemia can be taken immediately.
Cerebral ischemia is the leading cause of preventable death in cases of major trauma with severe traumatic brain injury (TBI).
The catheter is pre-calibrated during manufacturing, eliminating the need for zeroing while operating.
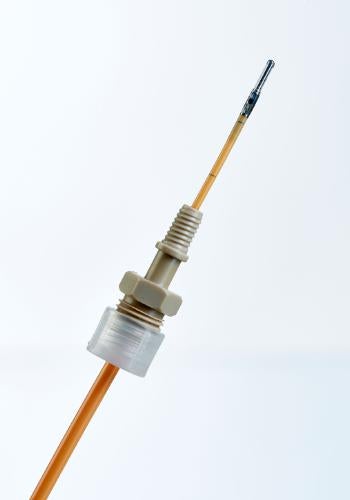
Tunneling Sleeve or low-profile PEEK BOLT is suitable in routing Neurovent-PTO catheter.
In addition, Neurovent-PTO has a sterility shelf life of two years and requires no refrigeration.
RAUMEDIC business unit director Robert Reichenberger said the company’s innovative neuromonitoring products have been sold in countries all around the world for over a decade to the benefit of patients and medical personnel.
“We are very pleased that we now have received 510(k) clearance from the FDA and can continue our market entry in the United States with the unique NEUROVENT-PTO,” Reichenberger said.
The addition of Neurovent-PTO places RAUMEDIC in the leading position for offering advanced neuromonitoring devices for managing critical care patients.
The company’s line of Neurovent products comprises microchip ventricular catheters which provide ICP and ICT monitoring with simultaneous CSF drainage and parenchyma catheters, which provide ICP and ICT monitoring.
All ICP and ICT monitoring catheters help save both time and money to medical centres as they can be directly connected to the patient’s bedside monitor.
According to GlobalData estimates, the neurological diagnostic equipment market was valued at $218m in 2012 and is expected to grow at a CAGR of 4.8% to reach $305m by 2019.
Image: Neurovent-PTO with low profile PEEK BOLT. Photo: courtesy of PRNewsFoto/RAUMEDIC AG.





