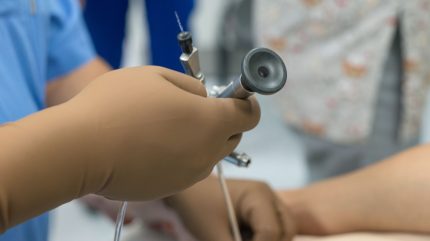
Boston Scientific has received 510(k) clearance from the US Food and Drug Administration (FDA) for its Asurys fluid management system, developed to provide irrigation and distention while carrying out endoscopic urologic procedures such as ureteroscopy.
The system aims to assist clinicians in maintaining optimal irrigation flow and controlling intrarenal pressure (IRP), both of which are important for procedures.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It is the latest addition to Boston Scientific’s StoneSmart Solutions product ecosystem for managing and treating kidney stones.
Asurys can be paired with the LithoVue Elite single-use digital flexible ureteroscope, which supports real-time IRP monitoring.
When connected to the LithoVue Elite system, the Asurys device allows physicians to control irrigation directly from the scope handle.
Activation of an on-demand flush feature through the ureteroscope enables repositioning of stones without manual irrigation or reliance on support staff.
The Asurys system can also be used in percutaneous nephrolithotomy (PCNL), cystoscopy, and benign prostatic hyperplasia (BPH) procedures.
Boston Scientific will begin limited market release of the device in the US in the coming days.
Boston Scientific Urology chief medical officer Dr Ronald Morton said: “Based on feedback gathered through our work with urologists, we know measuring and managing intrarenal pressure (IRP) while maintaining irrigation flow for visualisation is increasingly important.
“Until now, there has not been a system that can automatically respond to IRP data in real-time. The Asurys fluid management system can automatically regulate irrigation inflow to help minimise the potential of exceeding physician-selected IRP settings.”
Earlier this year, Boston Scientific reported a range of data highlighting the benefits of spinal cord stimulation (SCS) therapies within its neuromodulation portfolio.



