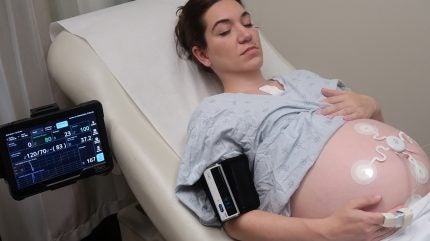
Sibel Health has received 510(k) clearance from the US Food and Drug Administration (FDA) for ANNE Maternal, a wireless maternal-foetal monitoring platform.
The platform is designed for simultaneous, continuous monitoring of both maternal and foetal health.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Combining wearable sensor technology and clinical decision support, ANNE Maternal supports a move towards making continuous maternal monitoring a standard of care worldwide.
The product is a sister platform to ANNE One, which is currently FDA-cleared for adolescent and adult monitoring in clinical and home settings.
ANNE Maternal utilises soft, flexible epidermal wearable sensors that transmit continuous maternal vital signs, including respiratory and heart rate, blood oxygen saturation, and skin temperature, alongside foetal heart rate and uterine contraction data.
Unlike traditional systems requiring women in labour to remain in beds with wired equipment, ANNE Maternal supports patient mobility without compromising clinical monitoring standards.
The platform provides both bedside and central monitoring via real-time visual and audio alarms. An integrated electronic modified early obstetric warning system (MEOWS) offers clinicians automated alerts when readings suggest evaluation is necessary.
Additionally, ANNE Maternal integrates with wireless point-of-care ultrasound, allowing a single interface for comprehensive perinatal care assessment.
It is designed to deliver continuous monitoring in intrapartum and postpartum scenarios.
Development was driven by a $17.5m grant from the Gates Foundation, supporting deployment in low-income and middle-income countries.
Sibel Health CEO and co-founder Steve Xu said: “What makes this platform truly unique is its dual mandate: it meets the rigorous clinical standards demanded by the FDA while also being designed for deployment in the world’s most resource-limited settings.”
Sibel Health also secured an additional $5m grant to incorporate future AI-driven features into the platform.
ANNE Maternal is already operational in Pakistan, Rwanda, Nigeria, and India through partnerships with the University of Edinburgh and the National Institute for Health and Care Research (NIHR) Global Health Research Unit on Global Surgery.



