The orthopaedic devices market suffered a major revenue loss during the pandemic. However, the market has gradually recovered and surpassed the pre-Covid level globally. According to GlobalData, the orthopaedic devices market is expected to reach nearly $50bn in 2023. Here are the top orthopaedic trends that have emerged and will continue in 2023.
Rising number of outpatient procedures
Covid-19 is accelerating the shift in the site of care, with more procedures, particularly total joint replacements, taking place in outpatient or ambulatory surgical centres (ASCs) to reduce hospital bed occupancy rates. The shift largely depends on the regulatory environment and reimbursement policies. As more types of orthopaedic procedures, such as complex spine and joint replacement surgeries, are removed from the Centers for Medicare and Medicaid Services (CMS) inpatient-only list in the US, both patients and surgeons will seek systems and facilities that deliver the health service at the best. On the manufacturers’ side, more orthopaedic instruments are tailored to meet the needs of ASCs, with smaller sizes and remotely controlled features. Additionally, companies such as Stryker have launched an ASC-focused business to deliver solutions specifically to optimise clinical and financial outcomes in the ASCs.
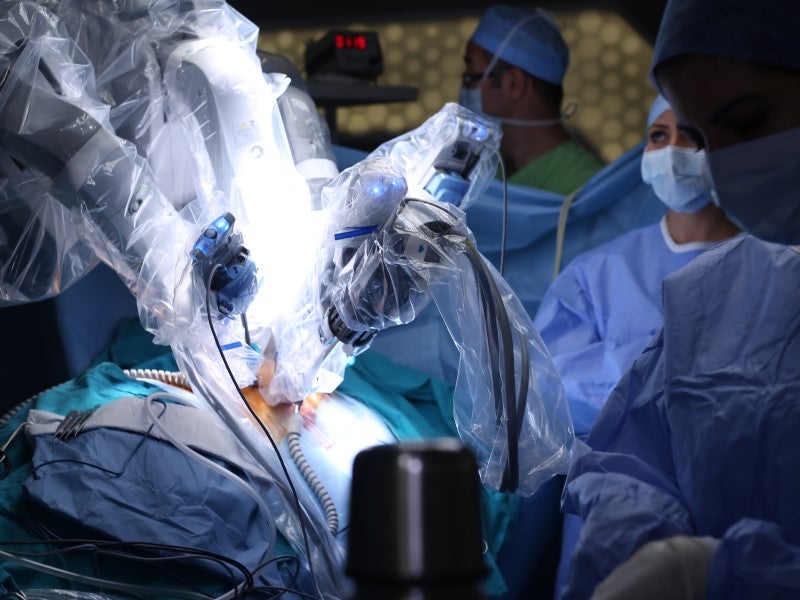
Rapid development of robotic surgery
Robotics-assisted surgeries will be more popular in the orthopaedic operating room in 2023. Orthopaedic robotic surgical systems are specialised to improve surgical outcomes for orthopaedic surgeries. They aid in various procedures, such as partial knee replacement, total knee replacement and total hip replacement, to ultimately help surgeons perform procedures more consistently and accurately than with manual techniques. Boosted by artificial intelligence (AI) and machine learning, new generations of orthopaedic robots are expected to further improve clinical outcomes. Unlike the general surgery robotics market – which is dominated by one company, Intuitive Surgical – all major orthopaedic manufacturers are competing in the orthopaedic robotics market. According to GlobalData analysis, the market for orthopaedic robotic surgical systems and accessories is expected to reach $984m globally in 2023, with year-over-year growth of 25.6%.
3D printing in orthopaedic implants
3D printing, or additive manufacturing, is the process of joining materials to make objects from three-dimensional data. As technologies’ development and costs come down, 3D printing is replacing traditional prototype development approaches across various industries, and medical usage is a fast-growing area. 3D printing has been used for custom-made orthopaedic devices, including joint replacements and artificial limbs. According to GlobalData analysis, the 3D-printed titanium implant Modulus from NuVasive is among the top five interbody products in the spinal fusion market. The revenue of the device has increased steadily since its launch in 2017. It has demonstrated faster revenue recovery than traditional interbody devices during since the start of the pandemic, indicating strong demand from healthcare facilities.
Regenerative medicine to treat sport injuries
Cartilage lesion is a common injury due to sports or trauma. Cartilage repair procedures aim to regenerate or replace articular cartilage to reduce joint pain and to prevent the development of arthritis. Third-generation or matrix-associated autologous chondrocyte implantation (ACI) was developed five years ago. For example, MACI from Vericel is the first US Food and Drug Administration (FDA)-approved product to offer autologous chondrocytes cultured on a porcine collagen membrane. It can be implanted with a generally simpler process than prior generations of ACI that required suturing a collagen patch. Researchers from the University of California, San Francisco, studied 2,010 patients who underwent ACI between 20120 and 2020. They found an increased rate of ACI from 2017-2019 compared to 2014-2016, whereas the re-operation rate was lower. Mirroring the increasing trend of ACI procedures, the company reported MACI revenue reaching approximately $130m in 2022, almost triple from its launch in 2017. GlobalData expects more regenerative medicine to be developed and marketed to meet the rising demand in 2023.
Supply chain challenges and inflation pressure
Prices of raw materials for medical devices have skyrocketed in recent years. The market has been facing a significant rise in transport costs and longer and more unpredictable delivery times. Price increases over the last two years are due to the effect of both the Covid-19 pandemic and Russo-Ukrainian war. Orthopaedic device manufacturers with more commoditised portfolios, such as hip and knee implants, will have less pricing power and a more limited ability to pass pricing on to hospitals. They are unwilling to increase prices and risk losing share to their competitors. Some companies may be unable to pass prices on to customers due to previous contracts. As costs remain high, brands that keep the same price will be suffering further margin erosion. GlobalData expects the trend to continue in 2023, as there is not a very clear path toward resolution of these issues for the orthopaedic industry.