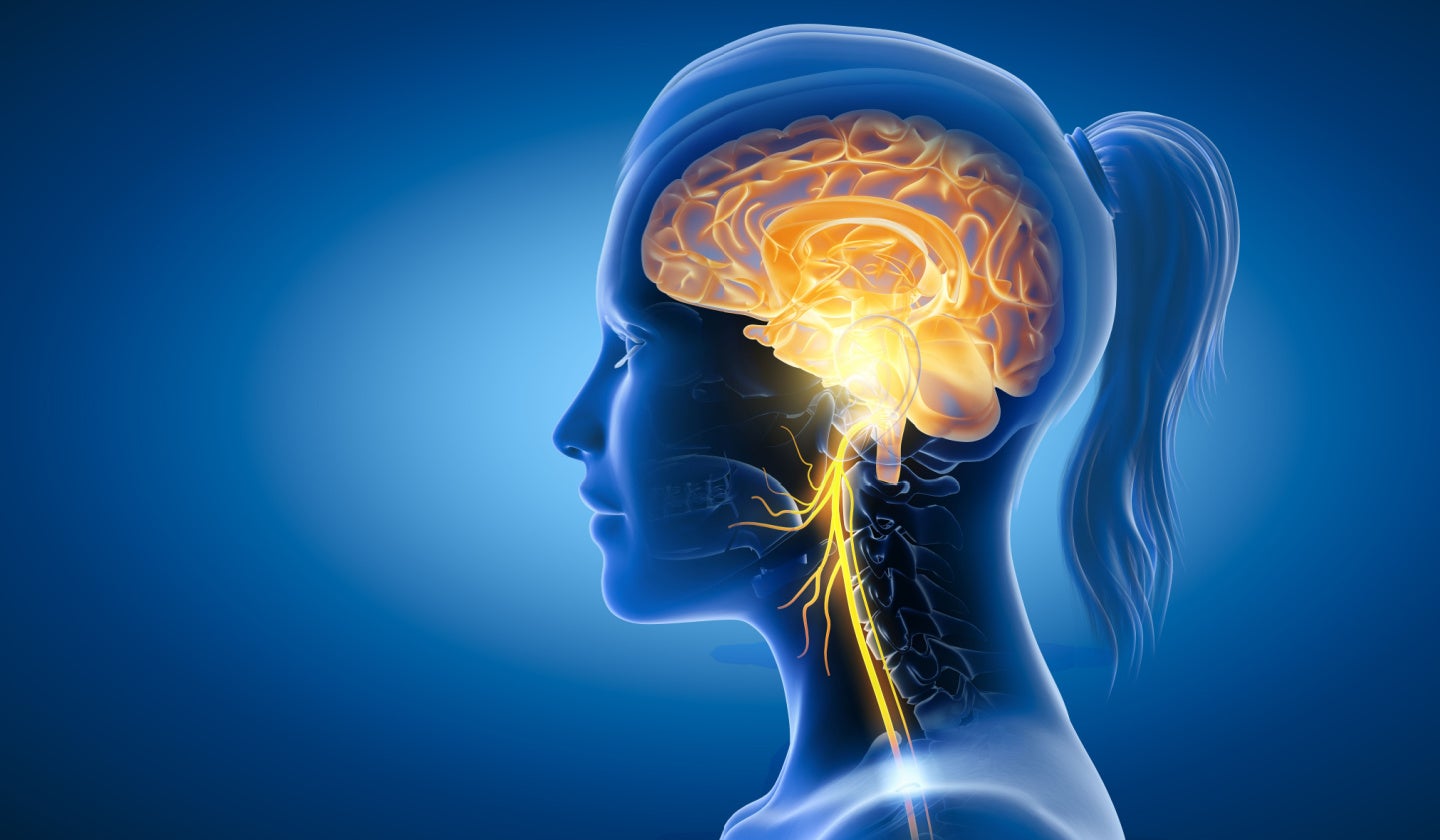
The vagus nerve acts as an information highway connecting the brain to a number of vital organs, helping to regulate bodily functions, send sensory information, control muscles and influence the immune system.
These pathways are not fully understood, but studies indicate that many are responsive to electrical stimulation. Pulse width, frequency and intensity can be adjusted to address specific neural pathways. This has resulted in multiple branches of neuromodulation research investigating different treatment pathways and uses for electrical vagus nerve stimulation (VNS) devices, particularly for difficult-to-treat conditions and patients unresponsive to conventional treatments.
Implantable VNS devices consist of a pulse generator that is surgically placed in the chest and a lead that delivers the electrical pulses to the vagus nerve in the neck. Implantable VNS devices have successfully treated drug-resistant epilepsy and treatment-resistant depression in more than 125,000 patients and have recently been approved for morbid obesity and stroke rehabilitation.
According to GlobalData, the implantable vagus nerve stimulators market is expected to grow from $679 million in 2022 to $1.27 billion by 2033. But existing implantable devices may be underutilised as they are costly, invasive, and often only used when all other treatment options have been exhausted.
New research into non-invasive VNS devices and new indications could allow for treatment without the need for surgery or implants. This means lower costs, reduced risks, improved accessibility, and better treatment access for more patients. Non-invasive VNS devices typically deliver stimulation through parts of the ear or neck by applying electrical stimulation through the skin. Transcutaneous electrical nerve stimulators are also under investigation for use in VNS therapies. Non-invasive devices may also lead to more robust and frequent research trials, as the need for surgery limits current clinical studies.
In Europe, the first approved non-invasive VNS device was Cerbomed’s NEMOS transcutaneous vagus nerve stimulator. This device was approved for the treatment of depression and epilepsy in 2010 and for pain in 2012. In the US, the first FDA-approved non-invasive vagus neurostimulator was electroCore’s gammaCore device in 2017 to treat cluster headache pain in adults.
Indications for these devices have since grown to include asthma, migraines, inflammation, sleep disorders, stroke and tinnitus. Recently, Tivic Health announced a study of a non-invasive bioelectronic device for vagus nerve stimulation using a novel approach. Tivic Health filed a patent for the device in July 2023 and has since received Institutional Review Board approval of its collaborative study with the Feinstein Institute’s Institute of Bioelectronic Medicine.
New VNS devices are increasing treatment pathways for indications such as depression and epilepsy. They are rapidly expanding into other cardiovascular, neurological, and systemic chronic health applications. Many of these indications are debilitating and/or life-threatening conditions that have exhausted all other treatment options, and patient-centred solutions are in high demand.
Neuromodulation is a multi-billion-dollar industry, and VNS is expected to continue growing as anatomical understanding improves and the need for cost-effective treatments for neurological conditions increases. Thus far, the VNS landscape is dominated by LivaNova, but as the breadth of indications expands and non-invasive technologies increase the potential patient pool, even smaller companies could achieve significant market penetration in the coming years.