The field of regenerative medicine has flourished in recent years due to advances in stem cell biology and tissue engineering. That trend continues to this day, this time thanks to an innovative bioengineering process designed by a team of researchers at the New York Stem Cell Foundation (NYSCF) (Sladkova et al., 2018).
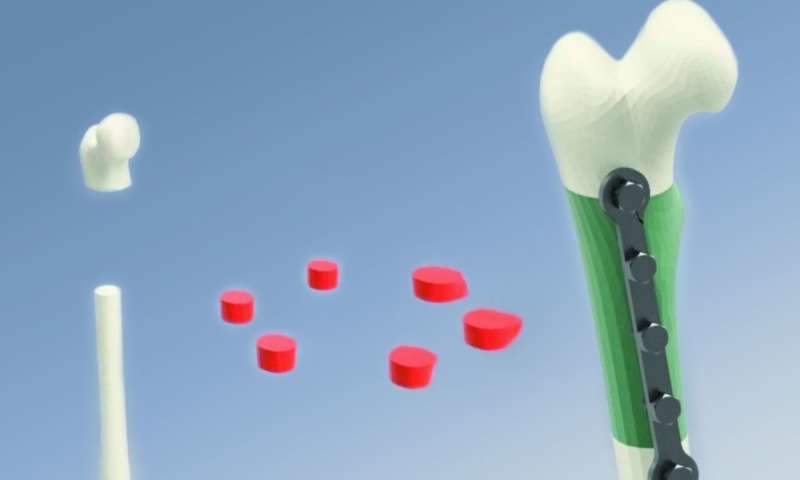
The Segmental Additive Tissue Engineering (SATE) process is used to produce bone grafts and leverages on the principle of “divide and conquer.” Digital reconstructions of the bone defect are segmented into different modules, each of which is defined by discoidal geometry and defined thickness.
This strategy of maintaining consistent spatial specifications in the smaller segments helps to fine-tune the seeding procedures and perfusion conditions in the bioreactor, and also helps to minimize product variability. Each of these modules are then used as a template for perfusion inserts and customized scaffolds, which are seeded with induced pluripotent stem cell-derived mesodermal progenitors (iPSC-MPs) and cultured in a modified bioreactor that helps to give rise to bone grafts.
These bone grafts are then introduced back to the patient’s body using compatible adhesives or fixation devices. All of these factors contribute to process optimization, a mandatory requirement for process reproducibility and translating the research from bench applications to clinical applications. The individual components and approaches used in this process serve to overcome the manufacturing-related challenges of producing graft materials for routine clinical applications.
Challenges for regenerative medicine
Regenerative medicine is often seen as a field that holds the promise of transforming human medicine, particularly for the treatment of diseases that have poor clinical outcomes with conventional drugs or medical procedures. The field includes cell therapies, gene therapies, and organ transplantations.
Challenges associated with the large-scale manufacture of these products appear at various levels, such as process validation and reproducibility. The new technique developed by the NYSCF team signifies a paradigm shift away from traditional engineering methods that aim to develop grafts from mesenchymal stem cells that use adult tissues as source of the bone-forming cells.
By using iPSC-MPs, the resulting stem cells have the potential to differentiate into all specialized cells that make up the bone tissues. The principle used in this innovative process may have a positive impact on manufacturing processes for other graft materials and help improve treatment outcomes in patients who undergo grafting procedures.