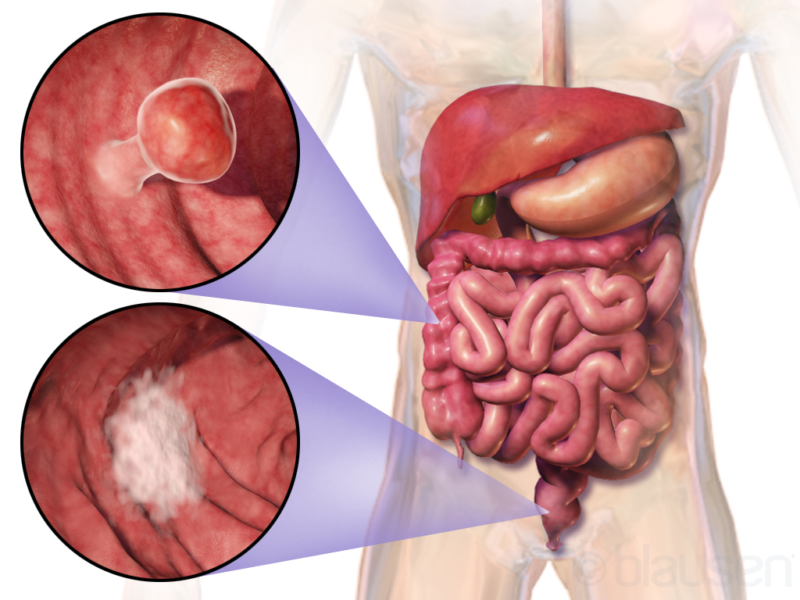
Colorectal cancer (CRC) is the third most common cancer occurring in men and the second most common cancer in women. In addition to inflicting devastating personal costs on patients, CRC places a sizable economic burden on national healthcare systems.
However, many people do not partake in recommended screening plans aimed at preventing this disease, with recent estimates projecting that just 67.3% of eligible US adults were up-to-date on their CRC screenings in 2016.
For average-risk individuals, many governments, cancer societies, and organisations recommend routine screening for CRC between the ages of 50–75 years. Options for CRC screening in average-risk individuals include stool or faeces-based tests performed every one to three years, or procedures such as colonoscopy performed every 10 years.
Traditionally, colonoscopy is considered to be the most accurate method for early detection of CRC. Despite this, studies suggest that compared to colonoscopy, the uptake of faeces tests is often better; this is possibly due to the easy-to-use and non-invasive nature of such tests.
Examples of popular in vitro faeces tests include the Guaiac Fecal Occult Blood Test (gFOBT) and Fecal Immunochemical Test (FIT); these tests are widely available and aim to detect small quantities of hidden blood in patient stool. The gFOBT and FIT tests have been on the market for decades. Of the two, studies suggest that FIT is the more sensitive and thus more popular assay.
In 2014, the FDA approved a new type of faeces test called Cologuard, which detects specific gene mutations and changes in DNA methylation known to be associated with CRC. Physician opinion is currently divided on which test to recommend, as Cologuard and FIT (or gFOBT) offer distinct advantages and disadvantages.
In average-risk individuals, the Cologuard, multi-target DNA test is more sensitive than FIT at detecting both CRC (92.3% versus 73.8%) and advanced precancerous lesions (42.4% versus 23.8%). However, the Cologuard test also generated more false positive results compared to FIT. Additionally, the Cologuard test needs to be taken less frequently than the gFOBT or FIT tests, but is currently significantly more expensive.
In addition to Cologuard, another non-invasive, test for CRC screening was also approved by the FDA in 2016, which measures the methylation status of the Septin 9 gene in blood samples. Ultimately, it is hoped that increasing the number of testing options available to patients will encourage greater participation in screening programmes, thus enabling CRC to either be prevented or to be caught early when it is most treatable.
Importantly, Medicare and most major US healthcare insurers now cover Cologuard. Earlier this year, the test’s manufacturer, Exact Sciences, announced that Cologuard has now been used to screen over one million patients. These sales figures, coupled with a strong advertising campaign and celebrity endorsements, indicate that Cologuard, and hopefully CRC screening in general, is currently gaining momentum.
Related Reports
GlobalData (2017). Feces Tests – RT & POC – In Vitro Diagnostics Market Analysis and Forecast Model, July 2017, GDME042MM
GlobalData (2017). Colorectal Cancer Diagnostic Tests – Medical Devices Pipeline Assessment, October 2017, GDME0454EPD
GlobalData (2018). Colorectal Cancer: Competitive Landscape to 2026, April 2018, GDHC006CL