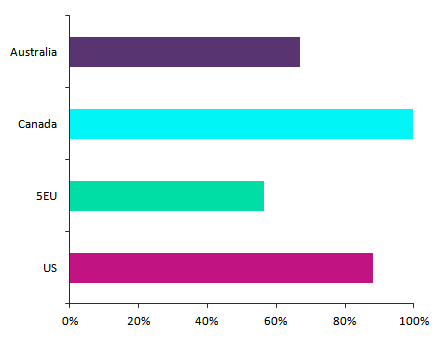If popular culture has taught us anything about anaphylaxis, it is that rapid injection of epinephrine is necessary to reverse the catastrophic symptoms and survive the episode. Though epinephrine itself has been off-patent for decades, the epinephrine market has remained a growing and competitive space based on the development of patented delivery devices known as epinephrine auto-injectors (EAIs), the most ubiquitous of these being Mylan’s EpiPen.
EAIs are designed to be portable and to quickly and accurately deliver a specific dose of epinephrine in the event of an accidental exposure to allergen. As part of an upcoming report on the treatment of peanut allergy, a disease for which anaphylaxis is a serious concern, GlobalData conducted a survey among high-prescribing physicians (primary care providers and allergists) from the US, the 5EU (France, Germany, Italy, Spain, and the UK), Canada and Australia. The survey results highlighted an important trend: physicians from the four geographies prescribe EAIs to their peanut allergy patients at starkly different rates.
Figure 1 shows EAI prescription rates across the four geographies covered in the survey. While physicians from the US and Canada prescribed epinephrine auto-injectors to 88% and 100% of their peanut allergy patients, respectively, physicians from Australia and the 5EU prescribed the devices to a much smaller percentage of patients.
Figure 1: rates of epinephrine auto-injector prescription to peanut allergy patients throughout 8mm

Source: GlobalData; Survey of high-prescribing physicians across the 8MM. 5EU = France, Germany, Italy, Spain, UK; 8MM = US, 5EU, Australia, Canada
Key opinion leader (KOL) insight suggests that much of this geographical difference is rooted in the ways physicians choose to treat two groups of patients in particular: the very young (between 1–3 years old) and those with a history of only ‘mild’ reactions.
An Australian KOL interviewed by GlobalData explained that for mild peanut allergy in very young kids, probably half of his department doesn’t prescribe EAIs. Though the prescription rate typically increases as patients get older and go to school, he noted that it can vary substantially from practice to practice. Factors driving differences in prescription rate include where a particular physician received their training, who is the head of their department, as well as the physician’s particular level of risk aversion.
In contrast, a KOL from Canada asserted it would be “basically malpractice” to not prescribe EAIs to all peanut allergy patients, irrespective of the severity of prior reactions. It is well described in the peanut allergy literature that a mild initial presentation of peanut allergy does not ensure that subsequent reactions will also be mild. A US KOL explained that the only patients he didn’t prescribe EAIs to were those that were confirmed to have a rare, mild form of peanut allergy called Oral Allergy Syndrome. Otherwise, he prescribed an EAI to everyone, even if their history had been of only mild reactions.
GlobalData’s survey data provide unique insight into the differing attitudes of physicians towards the prescription of epinephrine auto-injectors for patients with peanut allergy. A better understanding for how geography and local perception of the disease can alter prescription patterns will be critical to better predicting the dynamics of this budding market in the future.
Related reports
GlobalData (2018). OpportunityAnalyzer: Peanut Allergy – Opportunity Analysis and Forecasts to 2027, to be published