Myocardial infarction – commonly known as a heart attack – is caused by ruptured atherosclerotic plaques leading to flow-limiting blood clots in coronary arteries. Historically treated through coronary artery bypass graft procedures, they are now commonly treated through percutaneous coronary intervention (PCI). During a typical PCI, an intravascular balloon is dilated inside of the occlusion to restore normal blood flow, following which a stent is placed to keep the vessel open.
While coronary stent manufacturers continue to innovate in the field of traditional metallic stents, the technology had not progressed beyond drug-eluting metallic stents (DESs) for quite some time. There are several problems with DESs, mainly that they remain in the patient’s body for the rest of their lives, providing an embolic risk as well as preventing any more stents from being placed in the same area.
Absorb: dissolving prospects
Thus, there was considerable excitement when Abbott Laboratories launched the Absorb. Described as a ‘bioresorbable scaffold’, the device was a departure from standard metallic stents in that it dissolves into the body, preventing any chance of re-stenosis or other adverse events from a foreign object remaining in the body.
When originally launched, Abbott heavily marketed this new device as the next step in the world of PCI. Upon release, Abbott pursued an aggressive commercialisation strategy, particularly in Europe and America, in an attempt to gain market share. However, ambiguous or negative clinical results resulted in an extremely low clinical uptake.
The ABSORB II trial showed that patients who had an Absorb device implanted had a significantly higher rate of myocardial infarction post-implantation than patients who were treated with best-in-class DESs. The ABSORB III trial was a further blow, largely confirming the findings of the ABSORB II.
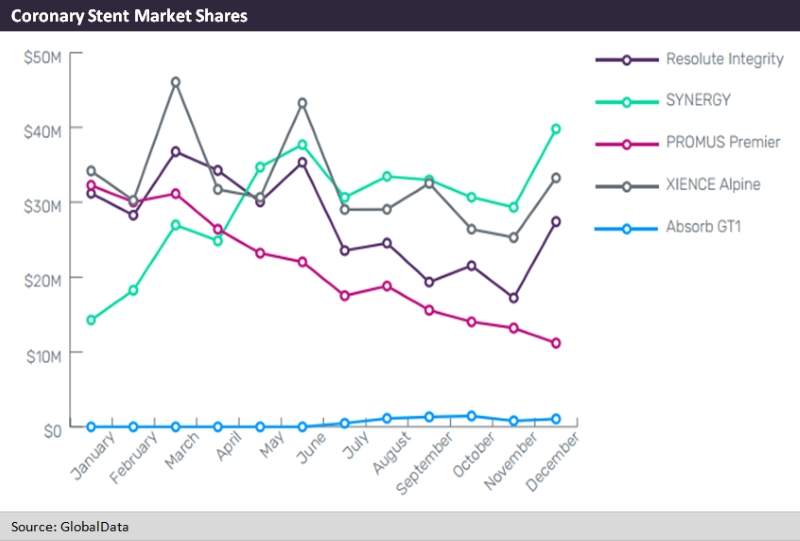
Due to these results, the Absorb seldom achieved more than 1–5% of the total market share, and Abbott ultimately shelved the product in September 2017, just over a year after it secured market approval.
Synergy soars

At the same time, Boston Scientific’s newest DES stent, the Synergy, secured 40% of the total US market share on the strength of its intra-procedural advantages, including its ease of deployment and trackability, or visibility in the fluoroscopes used by cardiologists to properly position the device in the patient’s vasculature. In fact, over the course of 2016 the Synergy overtook Abbott’s flagship DES, the Xience, previously the market leader in that segment.
These events serve as a cautionary tale: had Abbott focused on more incremental improvements to the Xience instead of gambling on unproven technology, it may have fared considerably better in the coronary stent market.