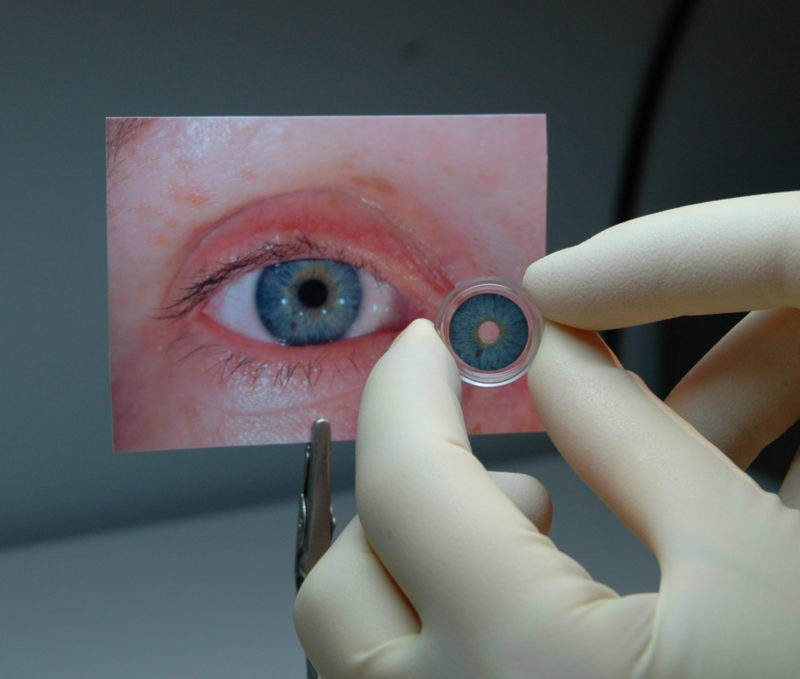
CustomFlex, by HumanOptics AG, is the first stand-alone prosthetic iris in the US, following its approval by the FDA.
The surgically implanted device will be available for adults and children whose iris is completely missing or damaged whether due to a congenital condition called aniridia or due to other damage to the eye.
A rare genetic disorder
Congenital aniridia is a rare genetic disorder in which the iris is completely or partially absent.
It affects approximately 1 in 50,000–100,000 people in the US.
The iris controls the amount of light entering the eye, and those with aniridia have sensitivity to light and other severe vision problems.
Reduces sensitivity to bright light and glare
In clinical trials, more than 70% of patients reported significant decreases in light sensitivity and glare as well as an improvement in health-related quality of life following the procedure, and 94% of patients were satisfied with the artificial iris’s appearance.
The CustomFlex Artificial Iris provides a novel method for treating iris defects that reduces sensitivity to bright light and glare.
It also improves the cosmetic appearance of the eye in patients with aniridia, offering patients an opportunity to improve how they see and how they are seen.