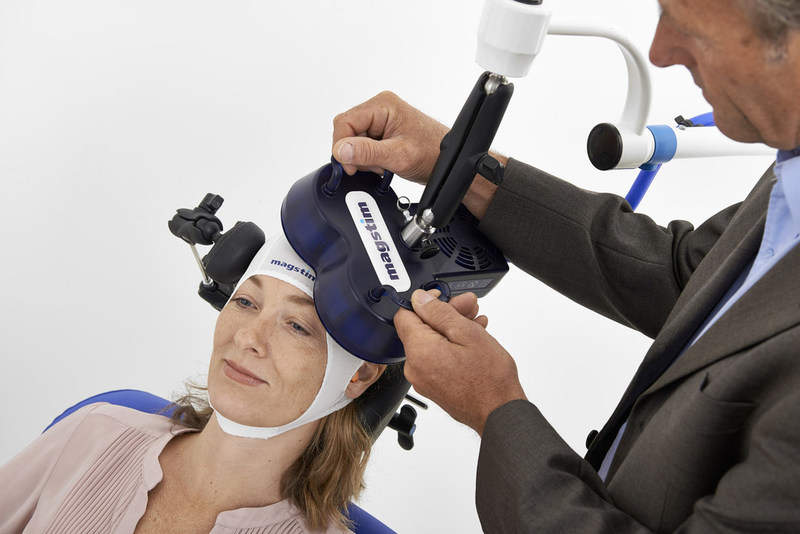
UK-based Magstim has launched its new Horizon transcranial magnetic stimulation (TMS) therapy to treat adults with major depressive disorder (MDD).
The technology platform is indicated for patients who could not achieve satisfactory improvement while using antidepressant drugs.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Claimed to be safe and cost-effective, the Horizon platform features a focused electromagnetic coil that quickly delivers a magnetic field to the target brain area.
The non-invasive, non-systemic TMS procedure is performed through emission of a series of repetitive, brief and focused magnetic pulses for activating brain cells and restoring brain activity level to normal.
Horizon includes an energy recovery system designed to provide easy, consistent stimulation while decreasing maintenance.
Magstim Group CEO Lothar Krinke said: “Horizon is Magstim’s next-generation technology platform for TMS applications.
“As we build on our proven record in research and continued expansion in the clinical market, we are confident that Horizon will form the basis for additional innovations that bring new hope to the millions who have been living with depression.”
The technology platform obtained CE-Mark as well as clearance from the US Food and Drug Administration (FDA) for treating patients who failed to improve from prior antidepressant medication in the current episode.
Magstim developed TMS as a research tool and optimised it for the treatment of nerve and brain conditions. The firm currently offers various research and clinical application products for different medical and research companies.





