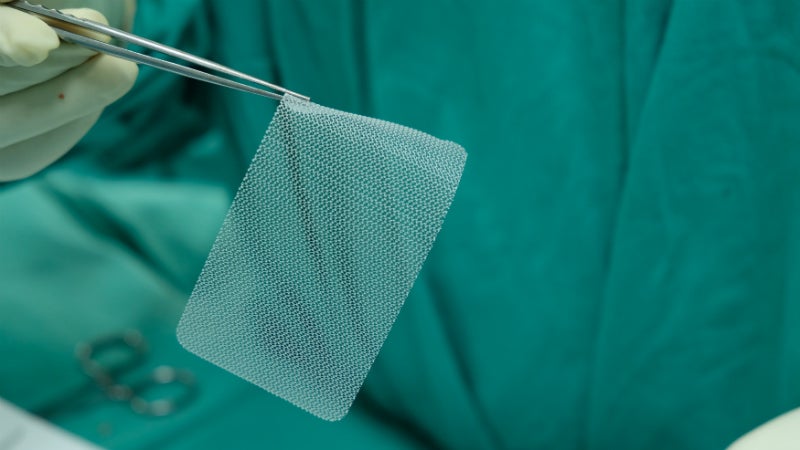
Hernia repair is one of the world’s most common surgical procedures.
A hernia occurs when body organs or fatty tissues protrude out of any wall or opening of the surrounding muscles. The most common site of hernia occurrence is the groin. Hernias are visible as bulges or lumps through the skin. They occur due to surgical wounds, weaknesses in the muscles, or birth defects.
One common way to repair hernias is with the use of meshes. A mesh is a surgical implant used to give extra support to the hernia-affected tissues. These meshes can be made from a variety of materials. GlobalData classifies these as synthetic meshes, composite meshes, or biological meshes. Synthetic meshes are made of synthetic materials such as polytetrafluoroethylene (PTFE), polypropylene (PP), or polyester (PET). These materials have high tensile strength and may be absorbable or non-absorbable depending on the material used.
Composite meshes are double-layered meshes with an absorbable side and a non-absorbable side. The absorbable side of the mesh is placed in contact with the hernia and allows tissue growth to strengthen the weakened abdominal wall, while the non-absorbable material prevents adhesion to the intestine. Biologic meshes are made from tissues of different animals such as cows (bovine pericardium) or pigs (porcine intestinal submucosal layer). These biological materials are high in collagen fibres and allow the native cells to populate and develop connective tissues, which allows for tissue growth in the weakened hernia.
While most hernia repair procedures are successful, it is estimated that about 10% of patients experience complications such as infections, pain, or organ damage. Since most hernia repair procedures are meant to be permanent, only a small number of these patients are eligible to have hernia meshes removed in a process that could result in further complications. Recently, experts have indicated that sufficient clinical testing is not always performed for hernia meshes. In some cases, meshes were only tested on animals for a few days, and therefore do not properly assess the potential long-term effects of mesh implants.
A report by the BBC found that over 100 different meshes are used by the UK’s National Health Service (NHS), but many of these devices did not undergo adequate clinical testing, with very few clinical trials having been conducted. As a result, the Medicines and Healthcare products Regulatory Agency (MHRA) has stated that it will be on alert for any new information about faulty hernia repair meshes and will promote new regulations to ensure the safety of these medical devices, including requiring manufacturers to provide clinical evidence demonstrating the safety and effectiveness for marketed products used by patients.

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalData