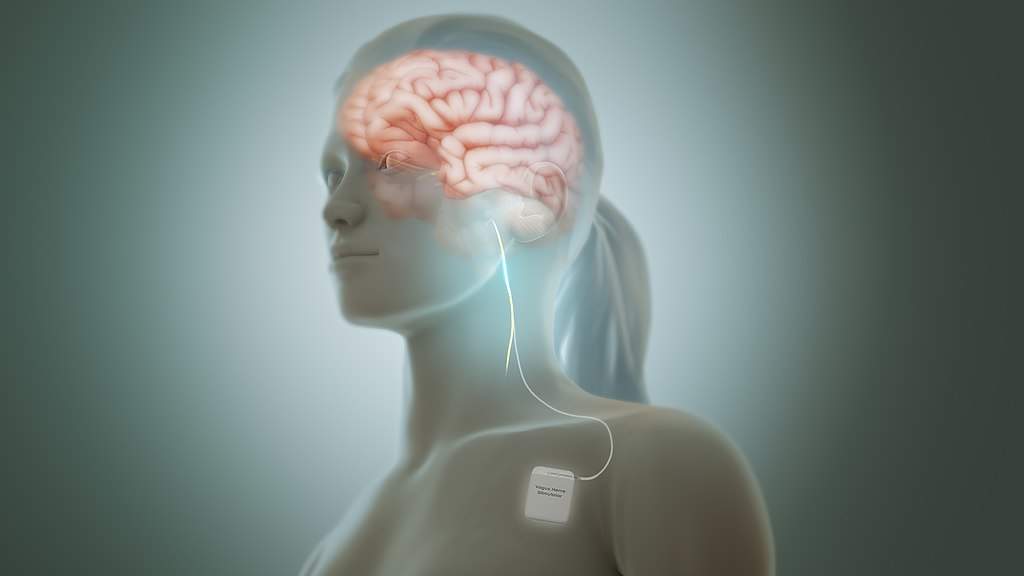
A new clinical trial at The Ohio State University is aiming to shorten the recovery period of motor skills for stroke patients via vagus nerve stimulation (VNS), drastically improving patients’ post-stroke outcomes and quality of life. Vivistim, a device developed by the Dallas-based company MicroTransponder, works by pairing VNS with muscle movement during rehabilitation. The pairing leads to strengthening of the neural circuits in the brain associated with motor function, learning, and memory.
As one of the leading causes of disability worldwide, acute ischemic stroke (AIS) is becoming increasingly prevalent due to both the growing aging population and an increase in risk factors such as high blood pressure, diabetes, high cholesterol, and obesity. Stroke, particularly in older adults, can lead to loss of motor function in multiple areas, such as movement, balance, and coordination; however, early treatment has been shown to reduce the chances of poor post-stroke outcome.
With the rising rate of stroke, focus has been placed on improving the prevention, intervention, and rehabilitation stages of stroke care. Neurovascular thrombectomy is positioned to be the new gold standard of treatment for stroke, as studies have shown that patients who received thrombectomy were “more likely to be functionally independent than those who received tPA [tissue plasminogen activator] alone” (Kulkarni, 2015). GlobalData expects the number of thrombectomy procedures to grow to 100,000 by 2027, which would lead to a decrease in post-stroke disability. Utilizing nerve stimulation for rehabilitation in combination with effective interventions would limit the impact of complications following an event.
The new application of nerve stimulation within stroke care could drastically improve the quality of life and recovery for stroke patients. If the Vivistim shows positive results in stroke patients regaining motor function earlier, many doors for potential rehabilitative treatment and reducing the burden of stroke on healthcare systems could be opened for the device.
For more insight and data, visit the GlobalData Report Store – Medical Device Network is part of GlobalData Plc.