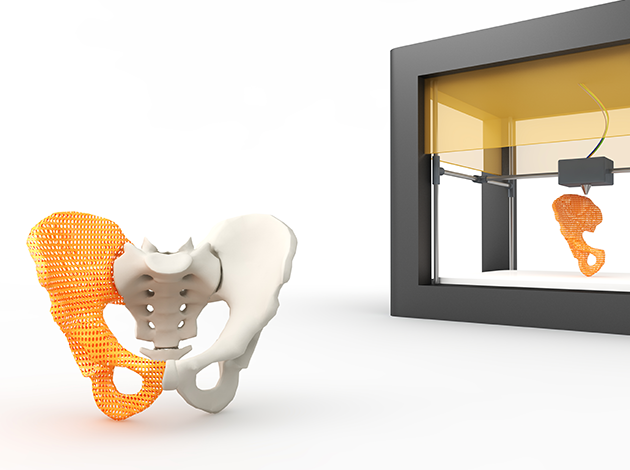
3D printing is a manufacturing process that creates a three-dimensional object layer by layer. The technique mostly relies on computer-aided design and manufacturing for custom-made devices. Current medical devices produced by 3D printing include orthopedic implants, dental restorations, and external prosthetics. In 2015, the first 3D printed pill was approved by the FDA. Current research focuses on leveraging the technology for bio printing, the 3D printing of human tissues by depositing cells layer by layer to grow organs.
The benefits offered by 3D printing are applicable for multiple stakeholders. Creating a customized implant for a patient based on their anatomy can translate into faster recovery time and fewer complications (Nuryyeva, 2016). A 3D printed model of a patient’s anatomy can help physicians plan elective surgical interventions, especially those that are complex. This can help optimize the procedure time and improve the quality of life for patients.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
There are a number of aspects that will affect the uptake of 3D printed medical devices; one is the regulations surrounding the technology. In 2016, the FDA issued a draft guidance for manufacturers that focuses on device design, manufacturing, and testing considerations when developing 3D printed devices. The regulations utilize the same pathways as those used for traditional medical devices for the approval of 3D printed devices. However, given the different working principles that define 3D printing, the regulations should take into consideration these differences and determine if these modifications will have an impact on the performance characteristics of the final device, such as strength, safety, and effectiveness (Reed Smith, 2015).
Qualifying for reimbursement is another factor that will affect uptake. In the US, 3D printed devices use the same premarket notification pathway as traditionally manufactured medical devices to enter the market. That means the reimbursement received for these devices will be the same, regardless of how these devices were manufactured. The absence of financial incentives for 3D printed medical devices can be a factor affecting the wider adoption of these devices in the market. This limitation can be offset by carrying out clinical trials and studies to establish positive outcomes with the use of 3D printed medical devices; the onus is on manufacturers in this space to lead this initiative.
Investments and innovations in the 3D printing space are other factors that can have a positive influence on the uptake of this technology for the medical devices sector. In 2016, Innovate UK, an initiative of the UK government intended to drive change within the UK’s manufacturing industry, set aside $5.5M to invest in projects that were set to improve the applications of 3D printing and aid in innovation. Such incentives foster an environment of healthy collaboration between companies and is a significant cornerstone for the development of advanced technologies.
3D printing is a technology that can positively transform the healthcare sector. The technology is rapidly evolving and expanding to include newer applications. However, it is also an innovation that needs to be clearly regulated. For the time being, it looks like manufacturers are in the lead in this space, leaving the regulations in need of catching up. To ensure the wider adoption of 3D printing in the market, it is essential that both the technology and the regulations are well established and developed to justify the position of 3D printing as the next industrial revolution within the healthcare space.





