
FDA approves marketing of 23andMe's genetic carrier test for Bloom syndrome
US Food and Drug Administration (FDA) granted approval for marketing 23andMe's Bloom Syndrome carrier test, a direct-to-consumer (DTC) genetic test to determine whether a healthy person has a variant in a gene that could lead to their offspring inheriting the disorder.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The company's Personal Genome Service 510(k) submission for this test report was assessed through the de novo regulatory pathway.
The FDA determined the company's submission did not have an applicable predicate device, and converted it to a de novo request. The agency took the further step to downclassify carrier status tests for autosomal recessive disorders.
Sorin and Cyberonics to create new medical technology company
Italian medical device firm Sorin Group and US-based Cyberonics agreed to merge to create a new medical technology company with a combined equity value of $2.7bn.
The proposed transaction, which was unanimously approved by the boards of directors of both companies, will see the two medical-device makers accelerate development of new products, and expand in areas such as cardiac surgery and neuromodulation.
As part of the deal, the two companies will combine under a newly formed holding company, NewCo. The combined company will also be a major player in cardiac rhythm management, especially in Europe and Japan.
Cerner takes over Siemens' health IT business for $1.3bn

US-based healthcare IT firm Cerner completed the acquisition of Germany-based Siemens Health Services' (SHS) assets, client relationships and associates for $1.3bn.
The acquisition will expand Cerner's annual R&D investment to $650m, and cumulative resources are expected to accelerate delivery of the company's next-generation of health IT solutions to its clients.
Cerner and SHS' former parent company Siemens are also moving forward with a strategic alliance to bring new solutions to market.
The alliance combines Cerner's health IT leadership with Siemens's medical device and imaging expertise.
As part of the alliance between Cerner and Siemens, each organisation will invest up to $50m during an initial three-year term, which will be used to integrate diagnostics and therapeutics into the electronic health record.
FDA approves Medtronic's PipelineFlex embolization device
US Food and Drug Administration (FDA) granted approval for Medtronic's Pipeline Flex embolisation device, developed for large and giant brain aneurysm treatment.
Available through a limited US launch in the next few weeks, the company's minimally-invasive flow diversion device for unruptured brain aneurysms is designed for more exact and controlled placement.
Baptist Health director of stroke and Cerebrovascular Center Dr Ricardo Hanel said: "Flow diversion has been a major breakthrough therapy for large or giant wide-necked brain aneurysms that are complex and have considerably higher risk of rupture and higher rates of complication with conventional treatment.
"With thousands of patients successfully treated with Pipeline embolisation device, the Pipeline Flex's innovative delivery system will result in further advancing endovascular treatment and care."
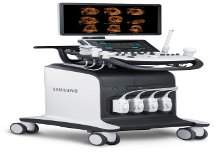
Samsung launches new ultrasound system WS80A with Elite performance package

Samsung Electronics America launched its new ultrasound product for women's healthcare, WS80A with Elite performance package.
The new system is designed to provide improved fetal heart imaging and diagnosis, as well as expanded 5D features including a new 5D Heart application.
Providing a workflow that can simplify examination of the fetal heart and improve reproducibility, the Samsung WS80A with Elite's 5D Heart tool is designed to generate nine standard fetal cardiac views simultaneously in a single template.
SpineGuard and Neuro France Implants to develop smart screw

SpineGuard, a producer of disposable medical devices, entered a co-development agreement to integrate its Dynamic Surgical Guidance (DSG) technology with Neuro France Implants' (NFI's) pedicle screw system.
The company said its DSG technology will be embedded into the pedicle screw system to offer accuracy, improve surgical workflow, and match health economic trends.
SpineGuard co-founder Pierre Jérôme said: "We are very pleased to partner with NFI on the development of this game-changing technology in a global annual market of one million pedicle screw-based procedures.
"Our R&D collaboration with Neuro France, initiated a few months ago, has already been fruitful with four labs, three generations of functional prototypes and very positive feedback from the surgeons involved in the pre-clinical evaluation."
Mitsubishi develops new multi-function irradiation nozzle for cancer treatment
Mitsubishi Electric developed a new multi-function irradiation nozzle for proton-type particle therapy systems for cancer treatment.
The nozzle, developed for particle therapy systems, will allow quick switching between broad-beam, layer-stacking and scanning particle beams.
It achieves flexible varied treatment matched to the tumour's individual location and shape to be performed in one treatment room.
BATM's diagnostic kit for HCV gets licence for sale in China
BATM Advanced Communications (BATM), a diagnostics business, received licence from China Food and Drug Administration to import, market and sell its diagnostic kit for hepatitis C virus (HCV) in China.
The diagnostic kit identifies by detecting antibodies to the virus using an ELISA screen.
BATM CEO Dr Zvi Marom said: "China is the world's largest market for hepatitis C diagnostics, and we believe that this approval will pave the way for the granting of licences for several more reagents that we have submitted for approval.
"Hepatitis C disease currently has no vaccine and it is estimated to kill approximately 350,000-500,000 every year, a number far greater than the recent tragedy spread by the Ebola virus."



