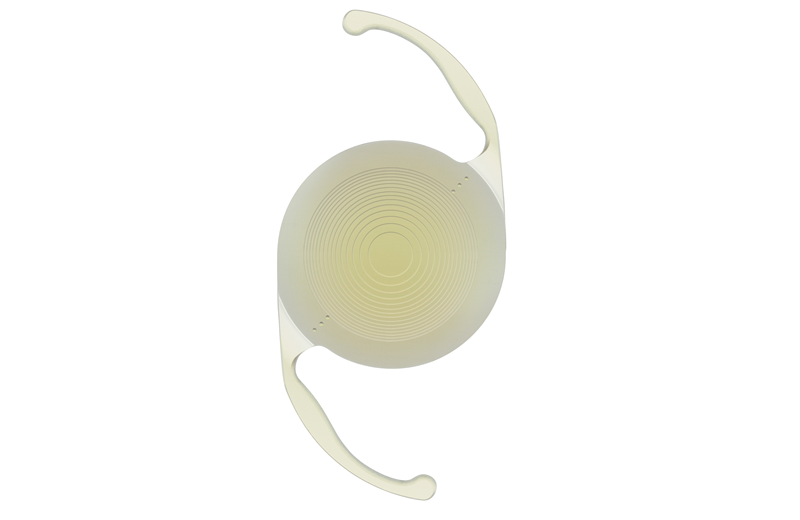
Eye care products company Alcon has commercially launched the AcrySof IQ PanOptix Trifocal Intraocular Lens (IOL) in the US for patients with cataracts, following approval from the US Food and Drug Administration (FDA).
PanOptix is currently being used to correct presbyopia in around 70 countries and has been designed to help the patient view in various lighting conditions.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The new lens uses ENLIGHTEN Optical Technology, which optimises intermediate vision without altering far and near vision.
Alcon CEO David Endicott said: “We are proud to bring this new class of IOLs to the largest eye care market in the world. We are leveraging Alcon’s years of experience with PanOptix in other countries to provide the best possible training and support for US surgeons.
“Our goal is to ensure optimal outcomes for cataract patients looking to correct their vision at all distances, with the vast majority of them never needing to wear glasses post-surgery.”
The approval followed a study in 12 investigational sites in the US. Patients who had the lens implanted had experienced clear vision.
PanOptix has been developed on the AcrySof IQ IOL platform, which has been implanted in over 120 million people in the world.
Currently, in the US, around four million cataract surgeries are performed per year. This number is expected to increase by 16% by 2024.
PanOptix and PanOptix Toric IOLs should be available early next year.
In April, Alcon separated from its parent company Novartis and began operating as a standalone, publicly traded entity.





