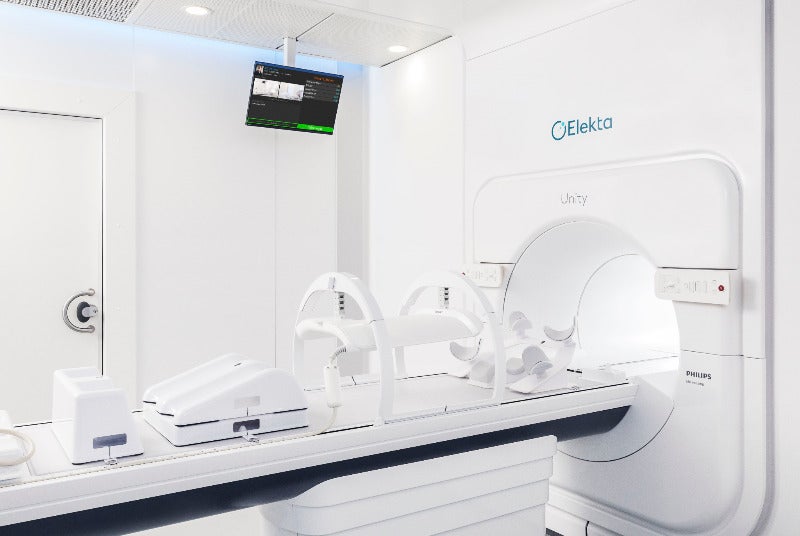
Precision radiation medicine solutions provider Elekta has received regulatory clearance from the Canadian Nuclear Safety Commission for clinical use of its Unity magnetic resonance radiation therapy (MR/RT) system.
Elekta Unity is a MR-linac intended to deliver personalised radiation therapy.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The device has been designed to adjust dosage depending on daily changes in the tumour’s shape, size, position and surrounding healthy anatomy.
Elekta noted that the MR/RT system’s real-time adaptive radiotherapy software allows accurate dose delivery with real-time tumour visualisation.
Unity already holds US Food and Drug Administration (FDA) clearance as well as the European CE-Mark.
Elekta president and CEO Richard Hausmann said: “In just over a year since its first clearance, Elekta Unity has enabled the transformation of cancer care in Europe, the US and Asia, and we are excited it is now available to Canadian patients.
“Unity is changing how leading cancer centres deliver radiation therapy, reshaping the dose in real time and targeting hard-to-treat cancers that previously could not be treated effectively with radiation.”
The Elekta Unity system features a 1.5T MRI scanner and a 7MV linear accelerator for precision beam delivery.
According to the company, the device facilitates management of hard-to-treat cancers and improves treatment options. Additionally, it enables assessment of tumour response with diagnostic imaging using the 1.5T MRI.
Sweden-based Elekta first unveiled the system at the 36th European Society for Radiotherapy and Oncology (ESTRO) meeting in Vienna, Austria in 2017.
Apart from the MR/RT system, the company’s portfolio includes products and solutions for stereotactic radiosurgery, oncology informatics, brachytherapy, neurosurgery and particle therapy.





