Embolic protection catheter company Emboline has announced the closing of a $5m bridge round of financing, ahead of an expected Series C funding round later this year.
The money will support Emboline in filing for CE Mark approval of its Emboliner embolic protection catheter, as well as preparing for commercial launch in Europe and a pivotal trial in the US.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The company’s recent SafePass 2 clinical trial showed that Emboliner collects five times the debris of commercial devices in transcatheter aortic valve replacement (TAVR) surgeries.
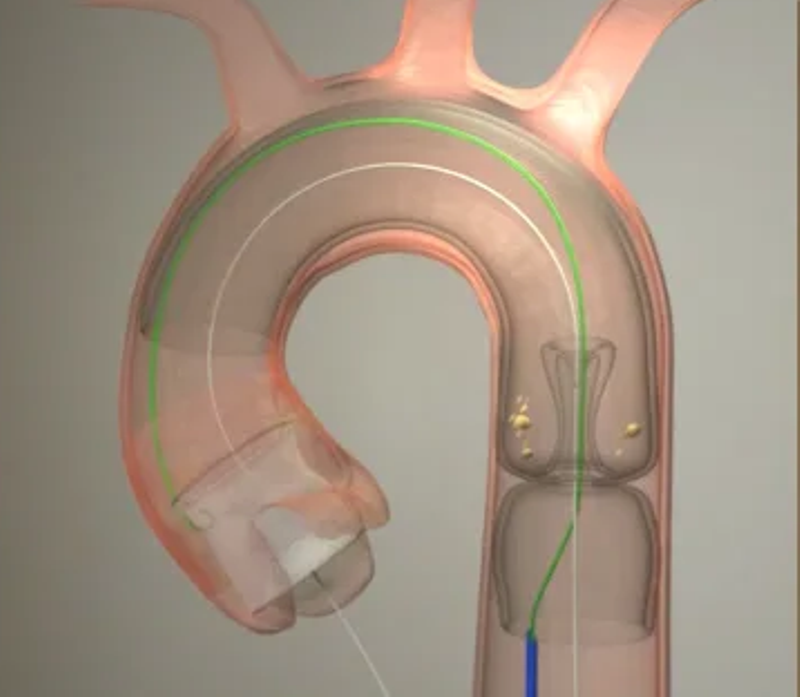
TAVR is a minimally invasive procedure to correct aortic valve stenosis, when a narrowed aortic valve fails to open properly. A catheter is inserted through the groin or chest and guided into the heart.
Emboliner is designed to provide total embolic protection of the brain and body during TAVR. A cylindrical nitinol mesh device covers the entire arterial bed and contains an expandable access port through which the TAVR devices are passed. This enables the device to capture and contain debris during the entire TAVR procedure.
Emboline requires no additional procedural access and minimal added procedural time.
Debris left over from TAVR procedures has been associated with a range of negative neurological outcomes for patients. These outcomes range from a major stroke rate of approximately 6%, overall stroke rates up to 22% and cerebral lesions in as many as 94% of patients.
Emboline founder and chairman Dr Amir Belson said: “It’s exciting to see that our concept of providing protection for both the brain and body is proving itself and demonstrating the potential to make the TAVR procedure even safer for patients. Our results in the clinic have validated the faith of our existing investors and triggered interest from a number of new investors who all strongly support the device and are eager to see it get it in the hands of the people who need it most.





