Health Canada has granted approval for the treatment of recently diagnosed and recurrent glioblastoma (GBM) using Optune from Novocure.
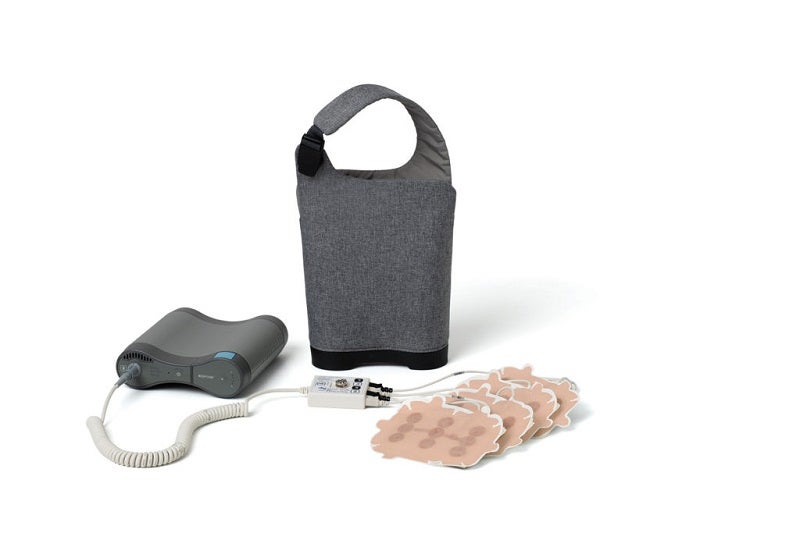
Optune is a wearable, portable medical device that creates Tumor Treating Fields (TTFields), which are electric fields for disrupting the division of cancer cells.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Using four adhesive patches called transducer arrays, it provides the TTFields therapy to the location of the tumour without increasing the chemotherapy-related side effects.
TTFields may destroy some of the cancer cells completely and have not been shown to affect healthy cells.
The small and lightweight Optune allows individuals to receive continuous treatment almost anywhere.
It is claimed to be the first treatment approved for GBM in Canada in more than 12 years.
Novocure Canada country manager Jovan Antunovic said: “We’re very happy that Health Canada has approved Optune for the treatment of GBM.
“We are grateful for the rapid and diligent review of our submission and for Health Canada’s approval of Optune.
“This is an important achievement in order to bring our therapy to more patients throughout Canada who can benefit.”
In the Phase III pivotal EF-14 trial, a combination of Optune and temozolomide was compared with temozolomide alone in 695 recently diagnosed GBM patients.
The trial participants treated with the Optune and temozolomide combination experienced 20.9 months of overall survival compared to 16 months for patients treated with temozolomide alone.
Mild to moderate skin irritation was the most common side effect related to Optune.
Novocure stated that Optune is available for GBM treatment in several countries in Asia, Europe and North America.
To date, more than 25,000 patients across the world have been treated with Optune.
According to the company, approximately 1,600 people a year are diagnosed with GBM in Canada.






