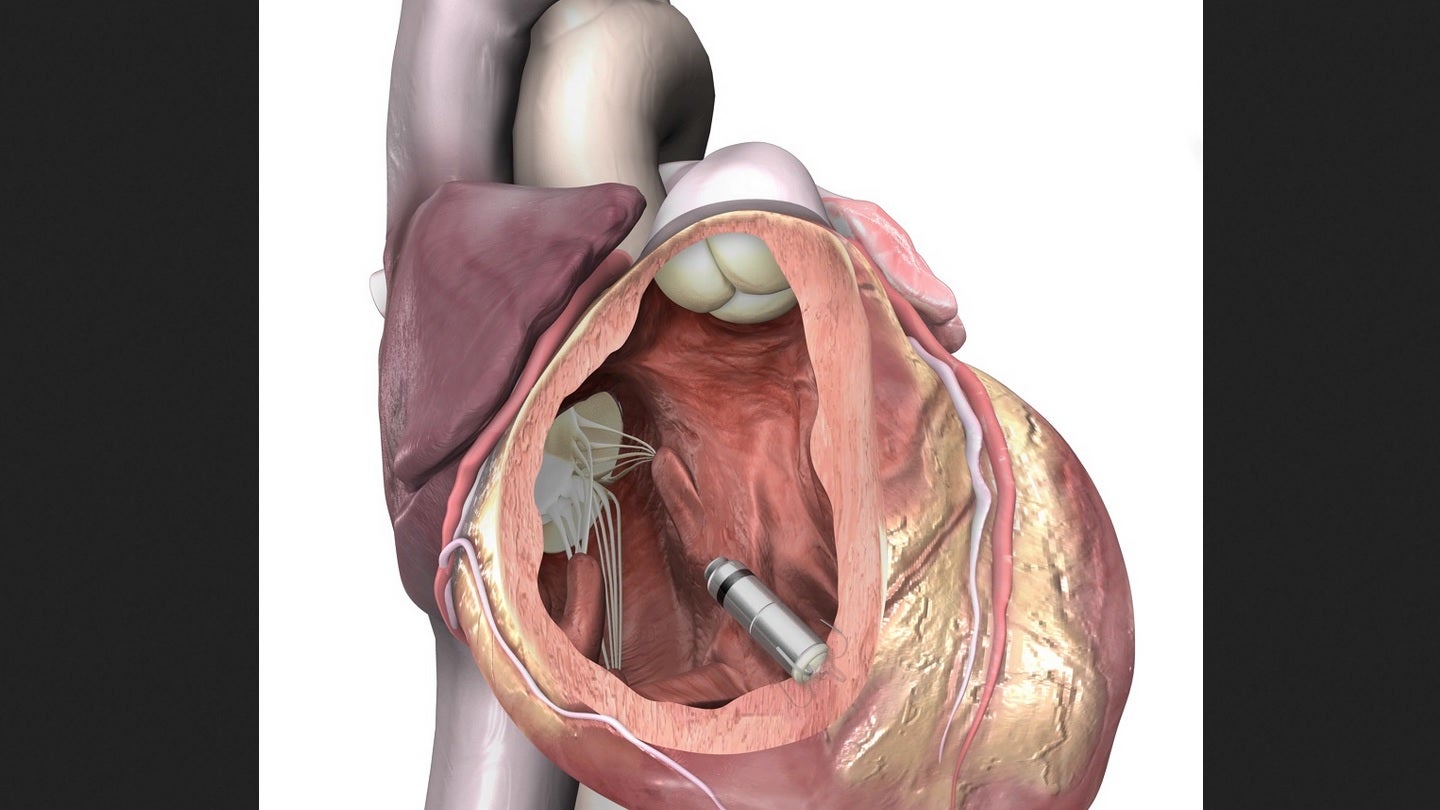
The US Food and Drug Administration (FDA) has granted approval for Medtronic’s next-generation miniaturised, leadless pacemakers called Micra AV2 and Micra VR2x.
Claimed to be the world’s smallest pacemakers, Micra AV2 and Micra VR2 offer 40% more battery life and easier programming compared to previous models in the Micra family of pacemakers.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Micra pacemakers are comparable in size to a multivitamin. They are claimed to be less than one-tenth the size of traditional pacemakers.
The Micra AV2 and Micra VR2 pacemakers can provide a battery life of 16 and 17 years, respectively.
They also provide reduced complications compared to traditional pacemakers.
Duke University Health System electrophysiologist Dr Camille Frazier-Mills said: “I’m excited to offer my patients the new Micra devices. This best-in-class technology transforms the patient experience by eliminating pocket-related complications and now reduces the chance that patients will need their device changed in the future.”
The new Micra AV2 is equipped with updated algorithms that automatically programme AV synchronous pacing, helping to coordinate the top (atrium) and bottom chambers (ventricle) of the heart.
It can also track faster heart rates in more active patients.
Medtronic Cardiovascular Portfolio’s cardiac pacing therapies general manager Dr Robert Kowal said: “Our goal is to improve the patient experience by continuously reinventing our groundbreaking leadless pacemaker.
“Since inventing the first battery-operated cardiac pacemakers 65 years ago, Medtronic has transformed pacing technologies to benefit patients, including the nearly 200,000 patients globally who have received a Micra device so far.”
Last month, Medtronic received approval from FDA for its insulin pump, the MiniMed 780G system.





