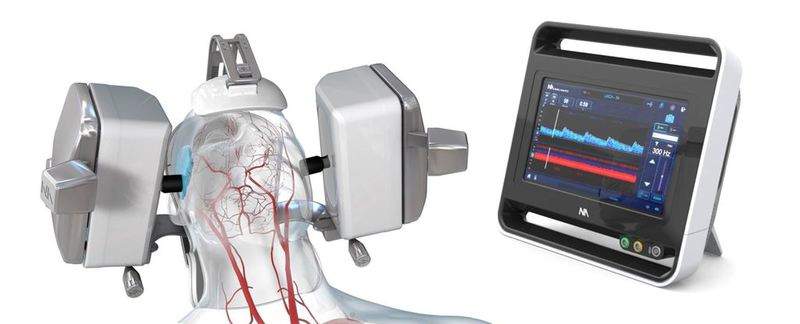
US-based medical robotics company Neural Analytics has received a CE Mark for its new robotic ultrasound solution called NeuralBot System.
The system is capable of automatically adjusting its position and orientation of its ultrasound products under professional guidance.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The NeuralBot System can be used with the Lucid M1 Transcranial Doppler Ultrasound System to monitor a patient’s brain blood flow characteristics, which can be utilised to diagnose multiple neurological disorders.
In April at the 23rd ESNCH Meeting in Prague, Neural Analytics demonstrated that there is no statistical difference between ultrasound blood flow data procured by NeuralBot System and data collected manually by an expert technician with conventional ultrasound platforms.
Neural Analytics co-founder and CEO Leo Petrossian said: “We are committed to advancing brain healthcare through transformative technology that empowers neurologists with the critical information needed to make clinical decisions and improve patient outcomes.
“Our products provide clinicians with a cost-effective and non-invasive assessment of a patient’s brain health, and can help clinicians diagnose brain disorders, potentially without the need for more invasive testing.”
University of Padua (Italy) Stroke Unit and Neurosonology Lab director Dr Claudio Baracchini stated that the technology will enable the medical experts to analyse blood flow characteristics and track emboli in patients.
Baracchini added: “It provides us with critical information on brain health in real-time to help us diagnose neurological disorders, prior to the need for additional, more invasive testing.”
Neural Analytics is planning to start the commercialisation of the NeuralBot System in combination with the Lucid M1 TCD System in Europe as the ‘Lucid Robotic System’.
The company has also secured 510(k) clearance from the Food and Drug Administration.





