
Arterys, a software-as-a-service (SAAS) analytics platform developer, has secured the US Food and Drug Administration (FDA) 510(k) clearance for its Arterys Cardio DL application to support cardiac MRI.
The software is based on a cloud computing and deep learning algorithm, which is a form of artificial intelligence.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
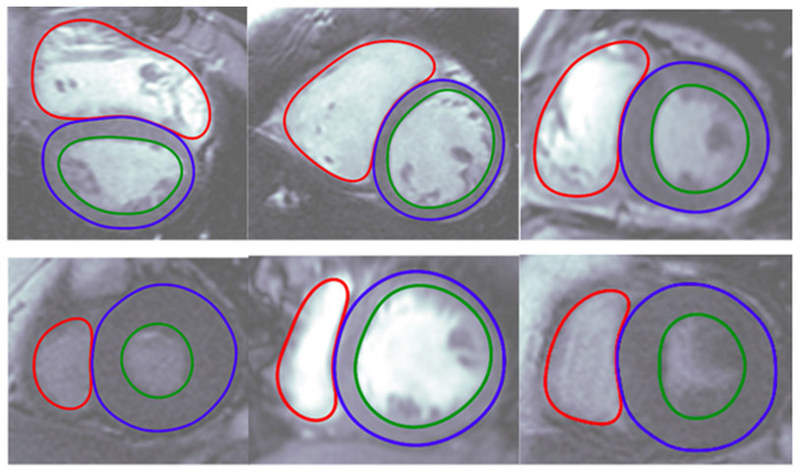
In a clinical setting, the algorithms provide automated, editable ventricle segmentations based on conventional cardiac MRI images, which are as accurate as the manual segmentations done by experienced physicians.
The automated contours generated by the Cardio DL display the interior and exterior of the heart ventricles.
Arterys CEO Fabien Beckers said: "Arterys is committed to broadly accelerate the transformation of data-driven medicine through advanced cloud medical imaging analytics applications while always protecting patient data privacy.
"Today's approval signifies a major regulatory milestone.
"This application demonstrates the power of deep learning combined with cloud supercomputation to aid physicians in interpreting medical images eliminating tedious manual tasks carried out on a workstation by accurately automating those processes."
Built on data derived from several cardiac cases, the vendor-agnostic Arterys Cardio DL application creates editable automated contours and paves the way for quick, precise and consistent ventricular function.
The trained deep learning algorithm of the software is a form of artificial intelligence demonstrated its efficacy in producing results within an expected error range comparable to that of an experienced clinical annotator.
Image: Automated contours generated by Arterys Cardio DL application. Photo: courtesy of Arterys.





