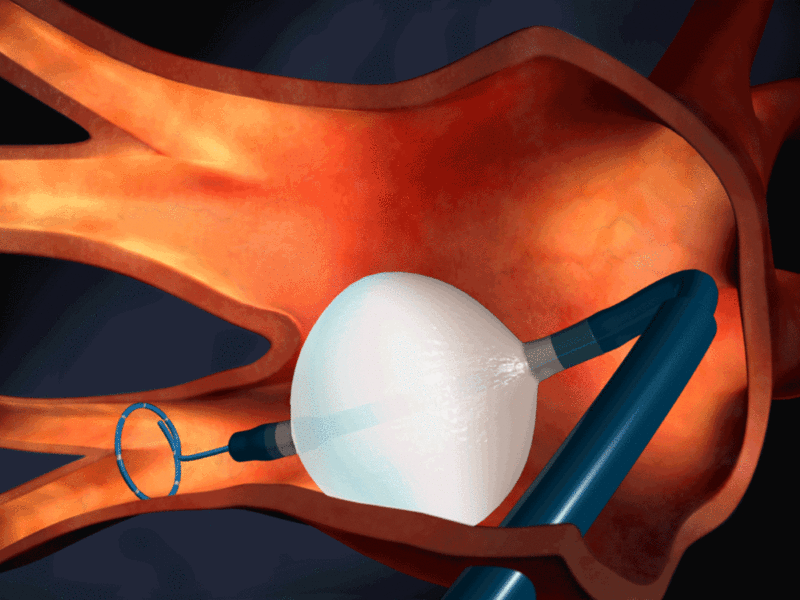

Medtronic has started enrolling patients in the STOP Persistent AF clinical trial of Arctic Front Advance cardiac cryoablation catheter for treatment of persistent atrial fibrillation (AF) in the US.
Equipped with the EvenCool Cryo technology, the Arctic Front Advance Cryoballoon is being developed to optimise delivery of coolant inside the balloon.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The prospective, multi-centre, single-arm STOP Persistent AF trial is designed to assess the safety and effectiveness of a pulmonary vein isolation-only (PVI) strategy using the catheter.
To be conducted at 25 centres in the US, Canada, Europe and Japan, the trial will enroll approximately 225 patients.
It will include a follow up period of 12 months after the initial cryoballoon ablation procedure.
Medtronic Cardiac and Vascular Group AF Solutions business vice-president and general manager Colleen Fowler said: "According to a study in JAMA, the number of patients with AF is expected to double in the next couple of decades and we want to ensure that physicians are equipped with the most innovative approaches and treatments.
"We hope this study leads to greater insights that can improve care for patients with persistent AF."
According to the trial's co-principal investigator Hugh Calkins, results from the trial are expected to aid in determining the possible treatment options for persistent AF patients.
The firm's cardiac cryoablation system has been used in more than 250,000 cases.
In the US, the system is approved to treat drug-refractory, recurrent, symptomatic paroxysmal AF and in Europe for the treatment of AF.
Image: Illustration of the Arctic Front Advance Cryoballoon placed in the pulmonary vein. Photo: courtesy of Medtronic.





