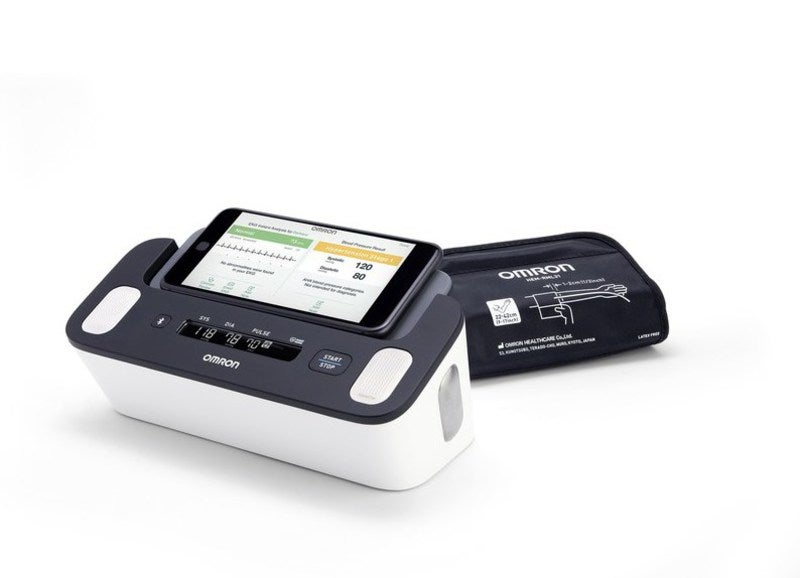
Omron Healthcare has launched a new FDA-approved blood pressure monitor, named Complete, with EKG capability.
Developed in alliance with AliveCor, the new device features an upper arm cuff for simultaneous monitoring of blood pressure and EKG readings at home.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
EKG is considered as an important measurement for patients with atrial fibrillation (AFib), a condition associated with a higher stroke risk.
By leveraging a new AliveCor-designed algorithm, Complete enables better detection of the possibility of AFib, in addition to medical-grade blood pressure measurement from Omron.
Omron Healthcare president and CEO Ranndy Kellogg said: “Complete is a groundbreaking tool for patients managing AFib and for those who suspect they may have an irregular heartbeat.
“Managing risk factors is essential for good heart health. By measuring more data points, we’re helping users and physicians gain a more complete heart health picture to evolve treatment and produce better health outcomes.”
The new blood pressure monitor can be connected with the Omron Connect mobile app meant for users to store, track data over time as well as share their heart health data with their physician.
Omron Healthcare key business unit head Tim Sheehan said: “Irregular heartbeat is difficult to diagnose because it’s just that – irregular. During our beta testing, Complete discovered my arrhythmia and, by using it with the Omron Connect app, I was able to share my readings with my doctor and discuss treatment. Now Complete helps me manage my condition.”
The company manufactures and distributes personal heart health and wellness products, including home blood pressure monitors and electrotherapy devices.
In January this year, Omron Healthcare introduced a wearable blood pressure monitor HeartGuide.






