UK-based diagnostics firm Owlstone Medical has entered a service agreement to provide its breath biopsy services to AstraZeneca for detecting new biomarkers associated with asthma and chronic obstructive pulmonary disease (COPD).
The companies will leverage Owlstone’s breathalyser to explore the applications of breath in distinguishing the phenotypes relevant for these respiratory diseases.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Breath Biopsy is a fast and non-invasive approach designed to identify breath-based biomarkers that can allow stratification and monitoring of patients.
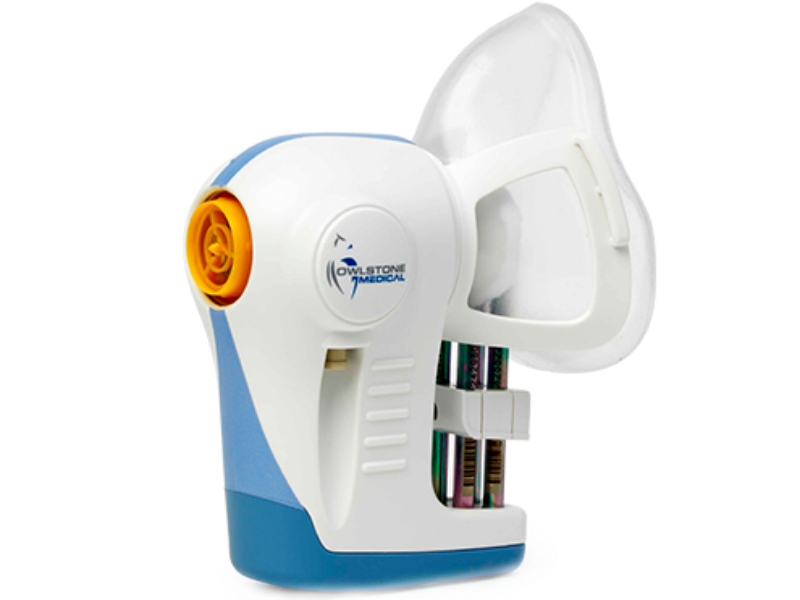
Owlstone’s platform works by detecting and analysing volatile organic compound (VOC) biomarkers in breath and its ReCIVA Breath Sampler device is designed to capture the VOC biomarkers in breath.
Under the terms of the deal, the company will also help AstraZeneca in developing classification algorithms for the identification of breath biomarkers that will pave way for precision treatments.
Owlstone Medical co-founder and CEO Billy Boyle said: “This agreement with AstraZeneca demonstrates our unrivalled expertise in breath biomarkers.
“Through our Breath Biopsy Services, we are well positioned to assist the AstraZeneca team to explore how breath biopsy can identify novel biomarkers for asthma and COPD and to optimise their application in precision medicine.”
The detection of disease phenotypes and treatable traits enable the study of the underlying heterogeneity of obstructive lung diseases. In turn, the information is expected to facilitate suitable treatment for the right patient at the right time.
According to statistics, about 334 million and 251 million people are affected with asthma and COPD, respectively, across the globe.
Similar to other chronic respiratory diseases, these conditions are considered as a substantial health and economic burden, leading to decreased quality of life and premature death.





