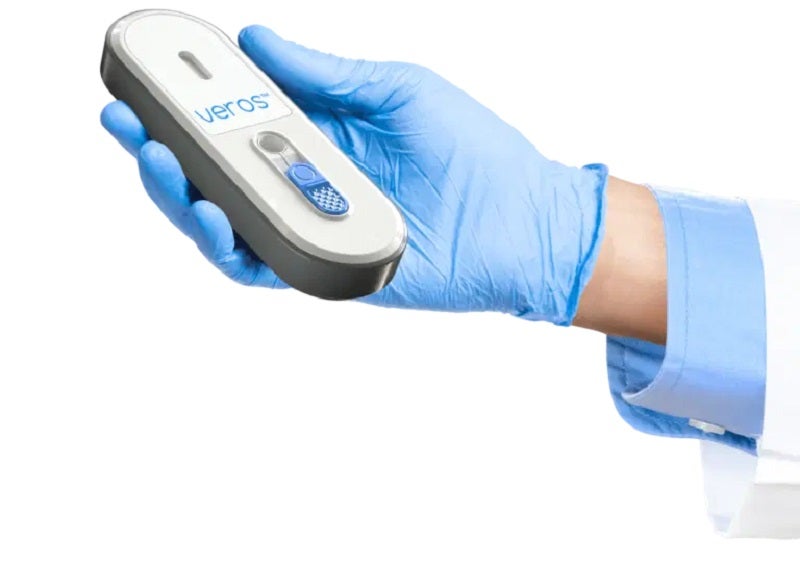
Sense Biodetection and Una Health have signed an agreement for the distribution of the Veros Covid-19 Test in the UK.
The point-of-care PCR quality Covid-19 test runs on the company’s single-use, self-contained, instrument-free Veros platform.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It uses rapid molecular amplification technology and provides instrument-free, PCR-quality laboratory results in 15 minutes without the need for an external power supply or reader.
The company stated that the platform helps to improve access to quick, point of care testing for many people.
Additionally, Veros helps reduce the spread of Covid-19 through precise diagnoses and improves treatment speeds.
Sense Biodetection chief commercial officer Ryan Roberts said: “Una’s strong relationships with customers in the point-of-care segment will be well served by the benefits that our rapid Veros Covid test can bring to the healthcare delivery process, providing the confidence of lab-quality results wherever they are needed.
“We look forward to working with Una as we introduce the first Veros-branded products to UK customers.”
The company stated that the deal with speciality medical distributor Una is the latest step in the commercialisation of the Veros platform, which awaits regulatory approval in both the US and UK.
The device is disposable and can be used at the point-of-care to overcome existing diagnostic testing barriers.
In 2019, Sense raised £12.3m in funding to develop and produce a range of instrument-free, point-of-care molecular diagnostic products.
Rapid molecular amplification technology is used in its molecular diagnostics tests to target infectious disease applications such as influenza.





