Medical technology company Stryker has closed the acquisition of US-based commercial-stage, medical device company Cerus Endovascular.
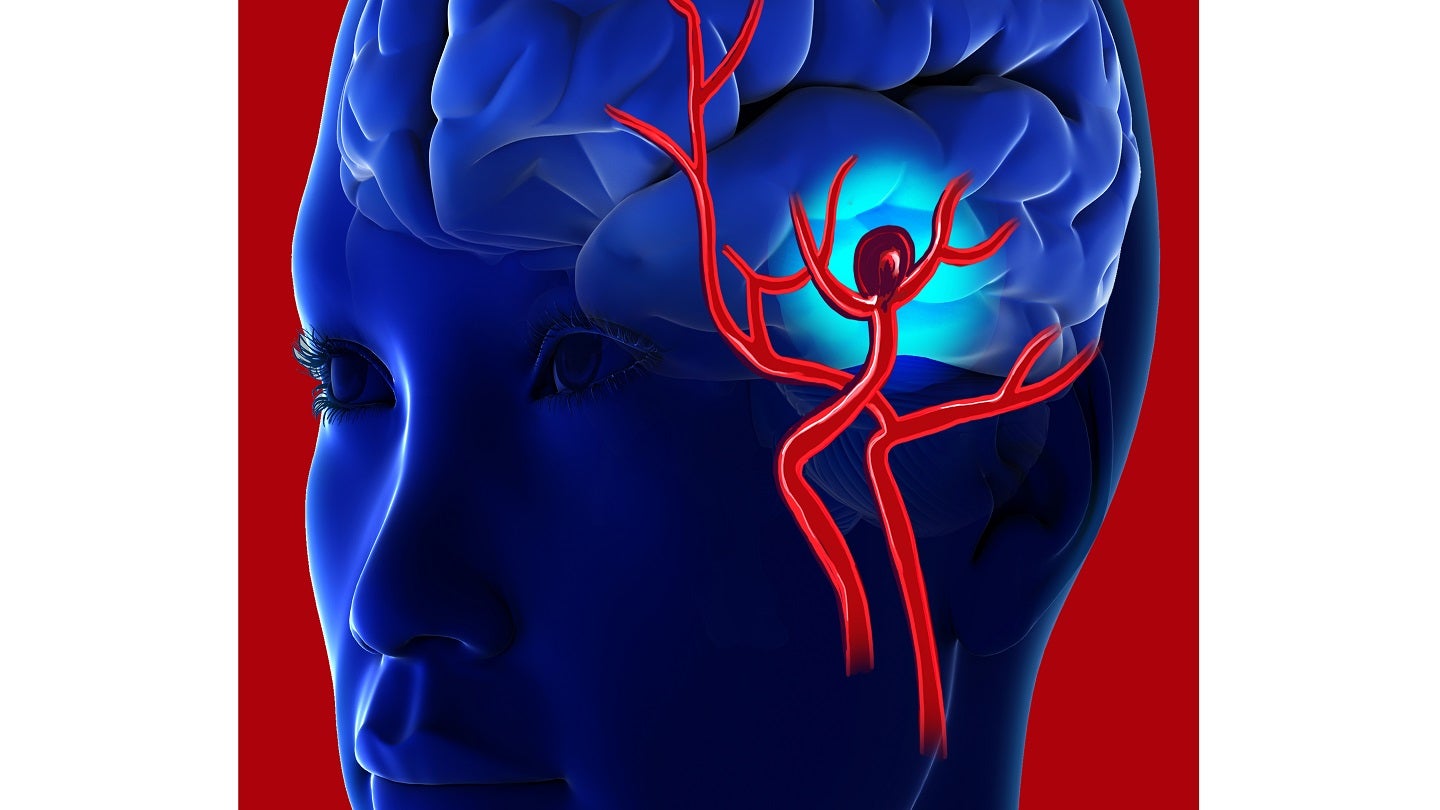
Cerus Endovascular is involved in the design, development, manufacture and supply of interventional neurovascular devices and delivery systems used by neuroradiologists for the treatment of intracranial aneurysms.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The devices are used in the treatment of acute, life-threatening neurological conditions.
The company’s offerings include the Contour Neurovascular System and the Neqstent Coil Assisted Flow Diverter. These products are CE-marked and are expected to expand Stryker’s existing portfolio of aneurysm treatment solutions.
Stryker’s neurovascular division president Jim Marucci said: “The acquisition of Cerus Endovascular is highly complementary to Stryker’s Neurovascular business, and strengthens our haemorrhagic portfolio globally.
“We look forward to working with the Cerus team to deliver on our mission of making healthcare better. Together, we will enable physicians to treat a broader range of aneurysms.”
Stryker’s Neurovascular division is focused on technology for the endovascular treatment of aneurysms. The addition of Cerus Endovascular’s portfolio is expected to accommodate the increasing demand for one-and-done intrasaccular aneurysm therapy.
The transaction came after a competitive auction process initiated by Cerus. Stryker and Cerus signed a definitive agreement for the deal in September 2022.
Global law firm Dechert was appointed by Cerus Endovascular to advise on the deal with Stryker.
UK regulatory body Competition and Markets Authority (CMA) reviewed the impact of the acquisition on competition in the UK market in February this year.
Last month, the CMA approved the acquisition, paving the way for the completion of the deal.
Stryker provides products and services in the areas of medical and surgical, neurotechnology, orthopaedics and spine.
In February this year, the company received 510(k) clearance from the US Food and Drug Administration for its Q Guidance System with Cranial Guidance Software for cranial applications.





