Medical device firm Subtle Medical has received the US Food and Drug Administration (FDA) 510(k) clearance and the European CE-Mark for its imaging technology, called SubtlePET.
The clearances allow marketing of SubtlePET in the US and the European Economic Area (EEA) regions.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
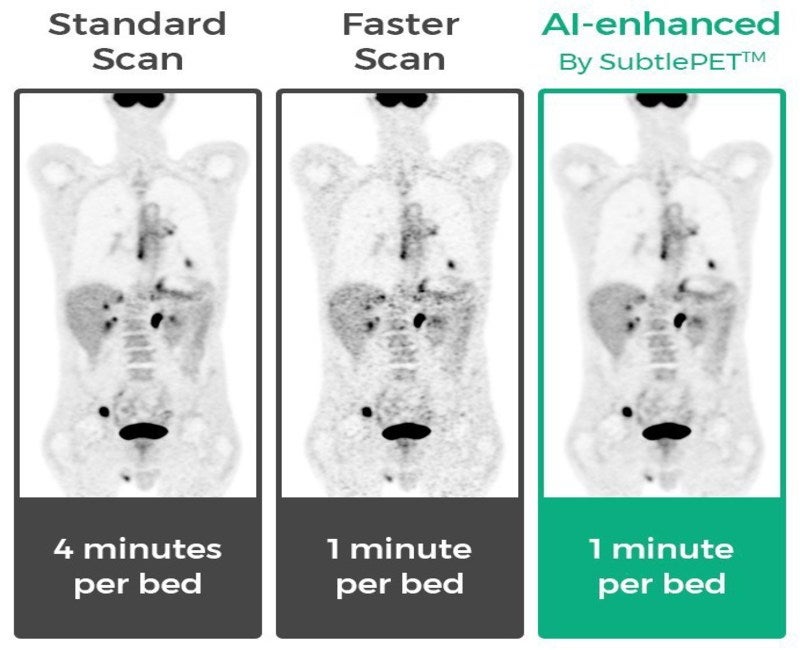
SubtlePET leverages artificial intelligence (AI) to enhance images from faster medical scans at hospitals and medical centres. The technology is said to offer better patient experience, improved exam throughput and provider profitability.
The company claims that the new technology allows completion of more exams in a day compared to traditional PET imaging, thereby reducing capital expenditures. It also minimises patient time in the scanner.
Subtle Medical founder and CEO Enhao Gong said: “Our focus on image acquisition and workflow differentiates us from other AI companies that are working on post-processing and computer-aided diagnosis products.
“We are not replacing radiologists–we are addressing the tremendous cost to US healthcare by leveraging deep learning in imaging at the infrastructure level to enable better and higher quality care.”
SubtlePET uses deep learning algorithms to enable use with any OEM scanner and PACS system in order to enhance images during acquisition, without disruption or alteration in the imaging workflow.
The AI technology also helps in improving the quality of noisy images from shorter scans, which is considered as an advantage for children and those undergoing repeat PET exams.
SubtlePET is in pilot clinical use at various US and international university hospitals and imaging centres.
Apart from SubtlePET, Subtle Medical is developing SubtleMR for use in imaging centres to speed up MRI scans and SubtleGAD to decrease gadolinium dosage during imaging procedures.





