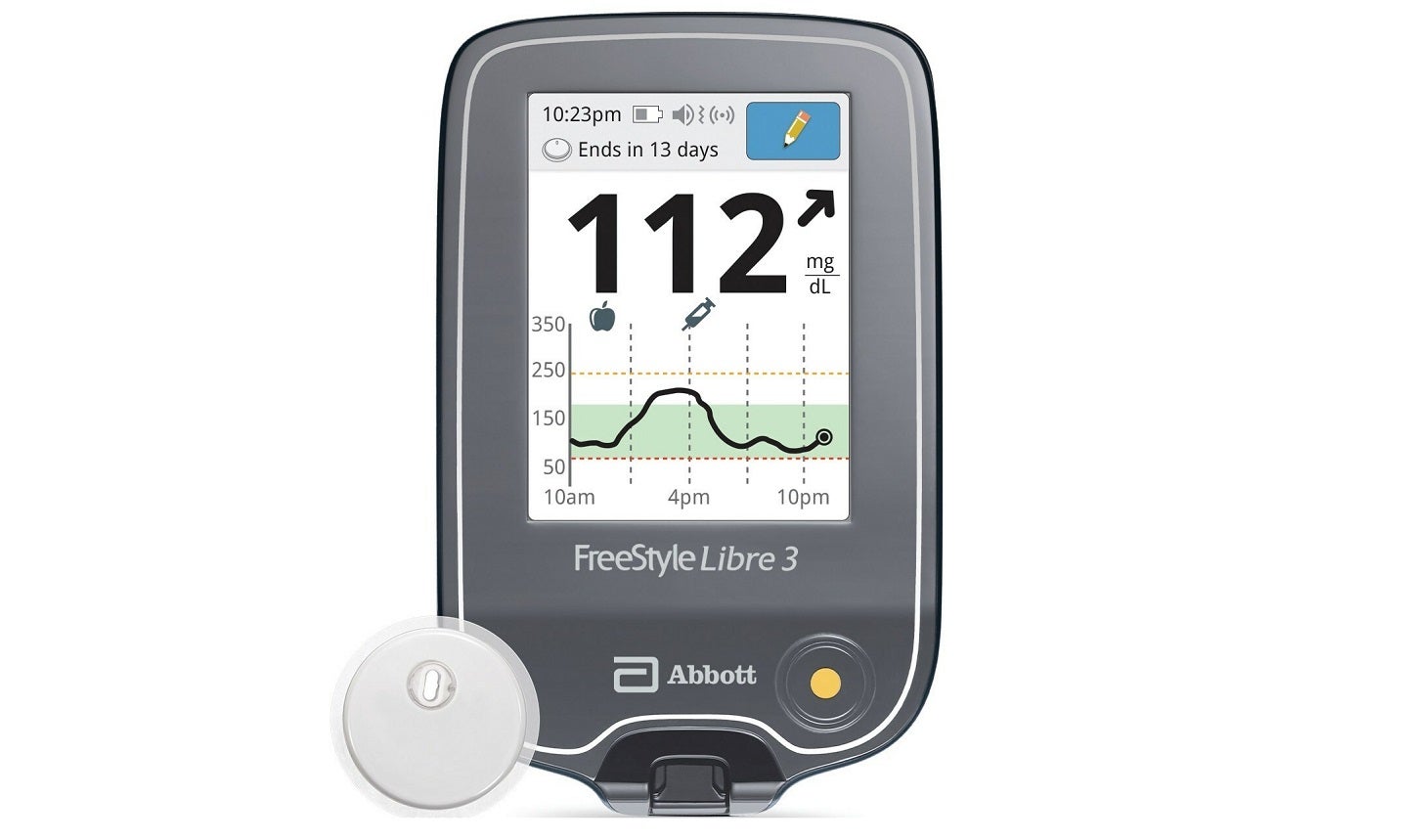
The US Food and Drug Administration (FDA) has approved a standalone reader device for Abbott’s FreeStyle Libre 3 integrated continuous glucose-monitoring system.
The new reader is a small handheld device and features a small sensor worn on the back of a person’s upper arm.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It displays real-time glucose readings on a large and easy-to-see screen and allows users to manage their diabetes easily and quickly.
The FreeStyle Libre 3 reader is equipped with a rechargeable lithium-ion battery similar to those used in mobile phones. Its user manual provides detailed information regarding storage, charging and usage as well as the use of the associated USB cable and power adapter.
Abbott stated that users of the FreeStyle Libre 3 system will have the option to check their glucose levels using the existing smartphone apps.
Abbott diabetes care business senior vice-president Jared Watkin said: “Our customers all over the world consistently tell us how our FreeStyle Libre technology has made an enormous, positive impact on their health and quality of life – they spend less time worrying and more time living.
“The FreeStyle Libre 3 reader provides more choice to people living with diabetes to have access to lifesaving technology that is smaller and easier to use and comes without the high-cost burdens of other systems.”
The company aims to get the FreeStyle Libre 3 system covered under the Medicare scheme to enable patients to get reimbursement.
Last month, the US FDA approved the company’s Epic Max stented tissue valve for the treatment of aortic valve disease.



