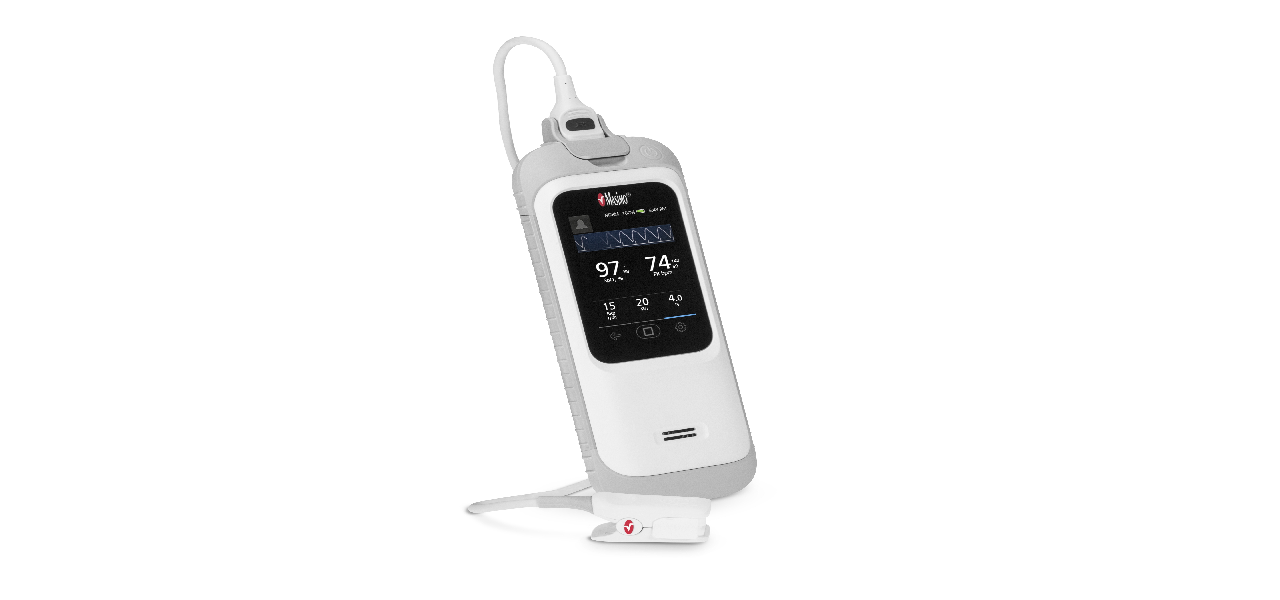
The US Food and Drug Administration (FDA) has cleared Masimo’s handheld pulse oximetry device called Rad-G Pulse Oximeter.
The rugged handheld device delivers clinically proven SET pulse oximetry, respiration rate from the pleth (RRp) and other key parameters for spot-checking and continuous monitoring.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It features a rechargeable battery, rubber casing, and a lightweight and convenient new direct-connect sensor that can be used for monitoring both adults and children.
The company said that clinicians will be able to quickly assess patients and make informed care decisions anywhere, using the pulse oximetry or vital signs checking provided by the Rad-G Pulse Oximeter.
The device can be used in a range of settings, including physicians’ offices, outpatient services, long-term care facilities, wellness clinics, first-response scenarios, and limited-resource environments.
Masimo founder and CEO Joe Kiani said: “With the Rad-G Pulse Oximeter, we set out to create an accessible, high-quality care solution that clinicians can rely on in a multitude of care settings. We are bringing our expertise in pulse oximetry to a smaller, more lightweight, rugged and versatile handheld device without sacrificing any of the advantages that help provide clinicians with critical insights into patient status.
“As the Covid-19 pandemic continues, it’s more imperative than ever that caregivers have access to the most accurate and reliable pulse oximetry monitoring technologies. We are proud to offer Rad-G among our suite of solutions powered by the breakthrough technology, SET pulse oximetry.”
Rad-G is compatible with Masimo’s portfolio of reusable and single-patient-use sensors. It is expected to provide clinicians with the ability to customise the solution based on the unique requirements of each care setting.
The company developed its first spot-check device in partnership with The Bill & Melinda Gates Foundation.
Last month, the FDA approved Masimo’s Pleth variability index (PVi) as a continuous, noninvasive, dynamic indicator of fluid responsiveness in select populations of mechanically ventilated adult patients.



