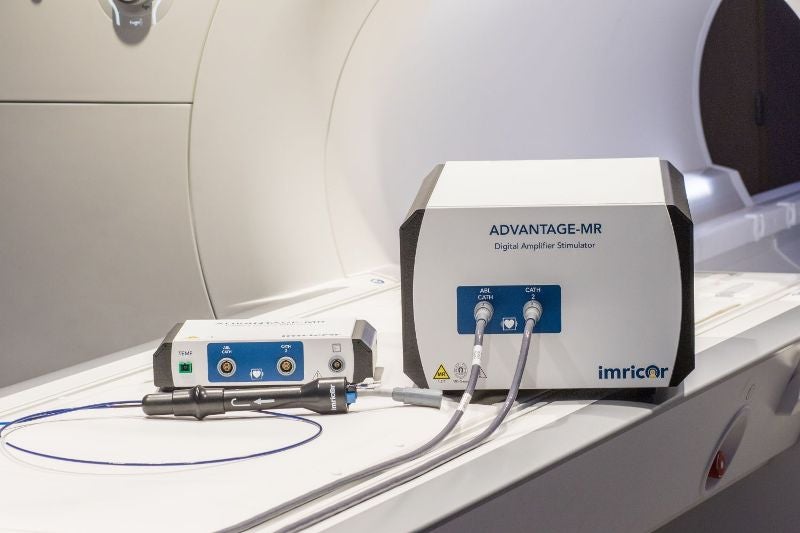
Imricor Medical Systems has secured CE mark approval for its Vision-MR Ablation Catheter and Vision-MR Dispersive Electrode for carrying out cardiac catheter ablations directed by real-time MRI.
The US-based medical device manufacturer stated that the CE mark approval of its catheter follows the previous endorsement of its Advantage-MR EP Recorder / Stimulator System in Europe.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Imricor claims to be the first company to provide cardiac ablation devices for use in the MRI environment.
Imricor chairman and CEO Steve Wedan said: “We are absolutely thrilled to bring to market our products for performing cardiac catheter ablations guided by real-time MRI.
“Receipt of CE mark approval marks a major accomplishment for the Imricor team, as we pursue our mission to improve the lives of people worldwide by delivering devices that enable iCMR procedures.”
“With inventory located in our European warehouse and a well-established roll out plan in place, we will now move swiftly to the execution of a controlled commercial launch to ensure smooth adoption and good clinical outcomes.”
Imricor has designed the catheter to function under real-time MRI guidance.
Imricor will provide hospitals and clinics with its products for interventional cardiac magnetic resonance imaging (iCMR) labs, where ablation procedures are performed using its catheter.
Imricor marketing director Nick Twohy said: “The future of cardiac ablation therapy is in MR-guidance and this product launch is the first step towards a new standard of care. We look forward to partnering with healthcare professionals to offer a new treatment option for patients who suffer from arrhythmias.”
The firm has signed agreements with several sites for the sale of its products. Featuring Advantage-MR systems, these sites are ready to begin procedures having secured the CE mark approval.





