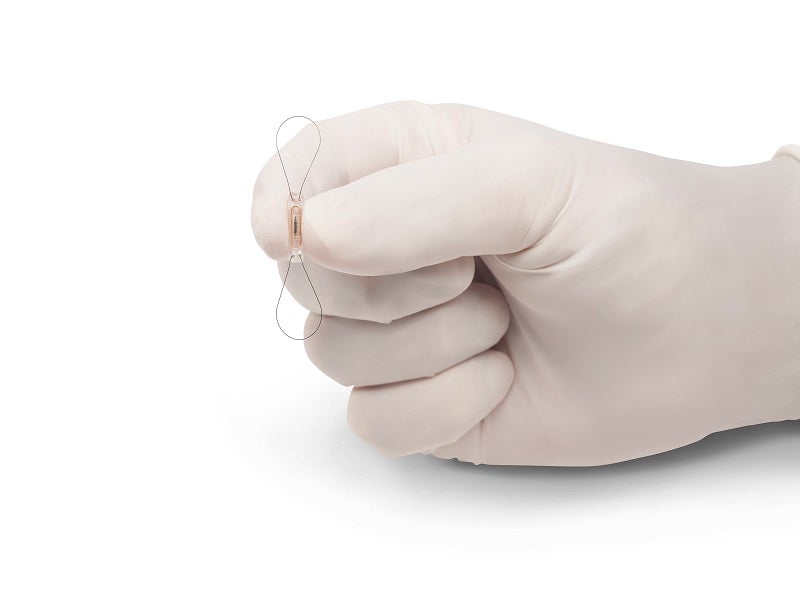
The CardioMEMS™HF system is used to measure changes in pulmonary artery (PA) pressure remotely, allowing physicians to begin or adjust therapy for heart failure (HF) based on the readings.
Developed by Abbott, the CardioMEMS HF system provides clinicians with information on PA pressure while the patient is at home, allowing them to remotely manage patients more proactively and reduce HF-related hospitalisations.
The system provides real-time patient change notifications, as well as simple and convenient access to protected data for individualised patient treatment. It also gives patients a greater understanding of the elements impacting their health, as well as a strong sense of control.
The CardioMEMS HF system can also be used for HF patients with preserved ejection fraction (HFpEF) or reduced ejection fraction (HFrEF). It helps to prevent the worsening of HF, lowering death rates and increasing quality of life for patients.
CardioMEMS HF regulatory approvals
The CardioMEMS HF system was first approved by the US Food and Drug Administration (FDA) in 2014 and by the European Union in 2016 for use in New York Heart Association (NYHA) Class III HF patients who had previously been hospitalised for HF.
The system received extended approval from the FDA in February 2022 for use in patients with Class II HF and those whose blood tests showed high levels of natriuretic peptides, indicating worsening HF.
The FDA approved the CardioMEMS HERO pulmonary artery (PA) pressure reader, a new component of the CardioMEMS HF system, in February 2026.
The CardioMEMS HF System is also commercially available in various countries in Europe and South America, as well as the UAE.
Heart failure
HF is a severe public health issue in the US, affecting more than five million individuals and resulting in over one million hospitalisations a year. More than 6.2 million Americans suffer from HF, with diagnoses expected to rise by 2030.
Elevated PA pressures can develop before signs and symptoms of HF decompensation and, therefore, offer a solid physiologic basis for HF patient care. Medical alternatives such as CardioMEMS can give patients and doctors actionable data to assist in the treatment of HF before it becomes more severe.
CardioMEMS HF system component details
The CardioMEMS HF system consists of a wireless sensor with a delivery catheter, CardioMEMS HERO PA pressure reader, patient or hospital electronics system, and patient database.
The CardioMEMS sensor is a paperclip-sized device that monitors PA pressure changes that signal worsening HF. The sensor is intended to be implanted permanently in the distal pulmonary artery using a safe right heart catheterisation surgery. It comprises a hermetically sealed capsule, an inductor coil and a pressure-sensitive capacitor.
The enhanced HERO reader, comparable in size to a laptop case, is designed for use with the CardioMEMS PA sensor. It supports a stable reading position for patients, helping to generate consistent daily PA pressure data for clinical teams. It incorporates both Wi‑Fi and cellular connectivity to simplify device setup.
The sensor collects non-invasive haemodynamic data, such as PA pressure waveform, heart rate, and systolic, diastolic and mean PA pressure. The data is measured using a hospital electronic system at the physician’s office, clinic or hospital.
Patients can also measure the data using a patient electronic system at their home, which is wirelessly communicated daily to their clinical team through a secure website that acts as the patient database such as Abbott’s Merlin.net Patient Care Network (PCN).
Physicians can adjust therapy or treatment based on the data to prevent progression to late-stage HF while also empowering patients to control their health virtually.
Merlin.net Patient Care Network (PCN)
Abbott’s Merlin.net PCN is a remote monitoring service that delivers PA pressure data to help patients receive more suitable treatment at an early stage.
The system incorporates remote PA pressure data with implanted electronic device (IED) diagnostics, such as atrial tachycardia and atrial fibrillation (AT/AF) load, ventricular pacing percentage, patient activity, day and night heart rate, and ventricular tachycardia/ventricular fibrillation (VT/VF) episodes with anti-tachycardia (ATP) and shock therapy.
GUIDE-HF clinical trial
The expanded indication of the CardioMEMS HF system was validated by results from the GUIDE-HF trial. The study included a randomised arm of 1,000 patients and a single arm of up to an additional 2,600 patients, with a total of 3,600 patients enrolled in 118 locations in North America.
The patients were implanted with a CardioMEMS sensor, although the treatment protocol for patients in the control arm was not guided by the device. The CardioMEMSHF system showed a significant 19% reduction in the study’s composite endpoint and 28% reduction in HF hospitalisations.
The trial demonstrated that patients with early-stage HF (Class II) benefited more from the device, with fewer hospitalisations, when their treatment was monitored remotely.
MONITOR-HF trial
The MONITOR-HF trial was conducted in Europe to evaluate health outcomes of using the CardioMEMS HF system in eligible patients with heart failure.
This prospective, randomised study included 348 patients across 25 centres in the Netherlands. All participants had New York Heart Association (NYHA) Class III heart failure and had either been admitted to a hospital for heart failure or had an urgent care visit requiring intravenous diuretics within the previous year.
Patients were randomly allocated to haemodynamic monitoring with the Abbott CardioMEMS device or to standard heart failure management with guideline-directed medical therapy (GDMT). Those who received the CardioMEMS implant were followed for at least one year. The complication rate was 2.3%, aligning with earlier studies.
Findings from the MONITOR-HF study showed a statistically meaningful 44% drop in heart failure-related hospital admissions among people with chronic heart failure receiving guideline-directed medical therapy (GDMT).
Participants also showed a clinically meaningful improvement in quality of life, with a mean seven-point increase in the Kansas City Cardiomyopathy Questionnaire (KCCQ) score, whereas scores declined in the control group, which did not receive the pressure sensor.
Furthermore, participants demonstrated a significant reduction in mean PA pressures at 12 months.