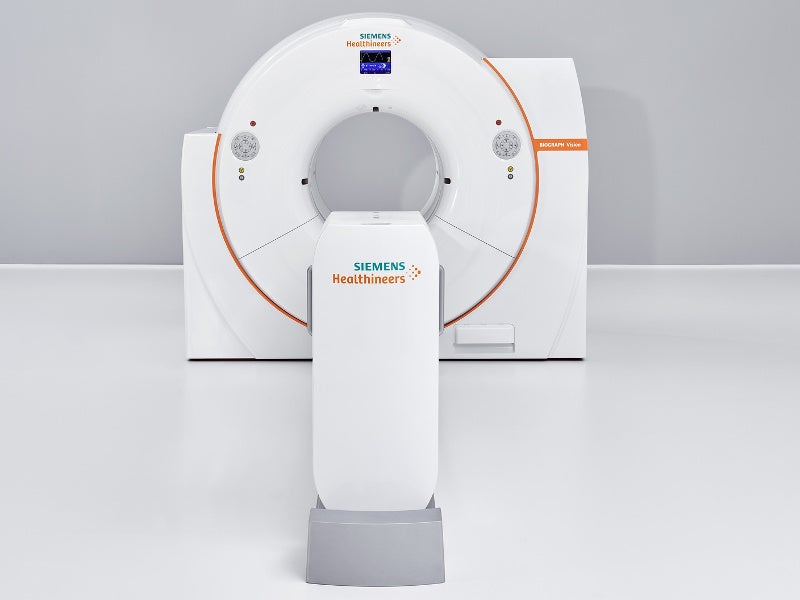
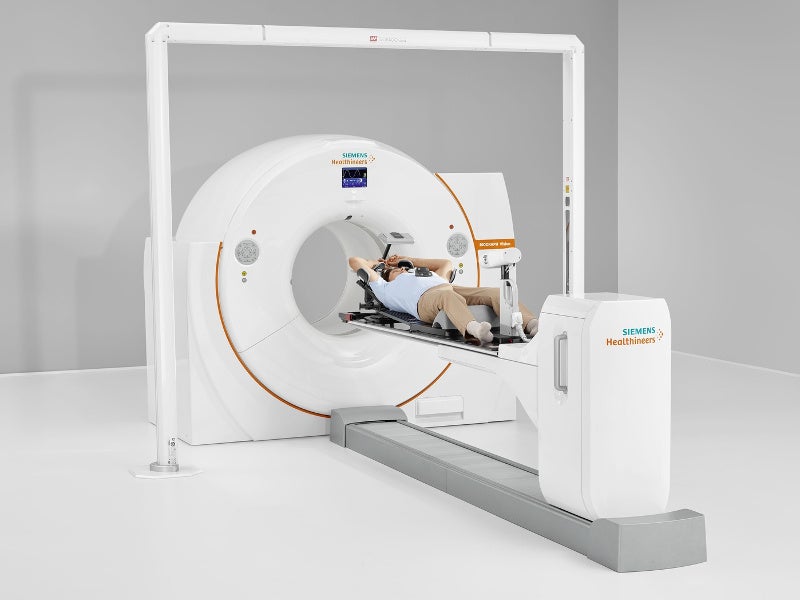
Biograph Vision™ is a first-of-its-kind, next-generation, high-precision combined positron emission tomography (PET) / computed tomography (CT) system developed by Siemens Healthineers. It is used for the whole-body examination of patients to diagnose cancer, as well as cardiac and nervous disorders.
The US Food and Drug Administration (FDA) provided 510(K) clearance to the device in June 2018. The RT Pro edition of the device was launched for comprehensive treatment planning with radiation therapy in October 2018.
The first Biograph Vision PET / CT system was installed at the Hospital of the University of Pennsylvania in Philadelphia, US, in February 2019, while the first system in the South-Asian region was deployed at the Apollo Hospital in Chennai, India, in May 2019.
Biograph Vision PET / CT system design and features
Biograph Vision PET / CT system measures 203.6cm-high, 234.4cm-wide and 136cm-deep. It features a wide 78cm diameter bore with a 136cm-long tunnel integrated with stereotactic and radiation therapy positioning accessories.
Patient bed with 127kg (500lbs) capacity is compliant with the American Association of Physicists in Medicine’s (AAPM) TG 66 guidelines.
PET and CT devices in the system function independently, allowing for single modality diagnostic imaging.
Details of the PET device
The PET device in the system features Optiso Ultra Dynamic Range (UDR) detector equipped with proprietary 3.2mm lutetium oxyorthosilicate (LSO) crystals and digital silicon photomultiplier (SiPM) sensors.
SiPMs cover 100% area of the LSO-crystal array, which allows for detection of the light emerging from the scintillation of LSO. It provides the fastest time-of-flight in the industry with the temporal resolution of 214 picoseconds and effective sensitivity at 100 counts per second (cps) / kBq.
The axial field of view of the device is 26.3cm, while the noise effective counts (NEC) are 1,870kcps.
Biograph Vision system CT device details
CT device incorporates SOMATOM Definition AS / AS+ and SOMATOM Definition Edge subcomponent.
The component reconstructs the X-ray transmission data taken from different angles and generates cross-sectional images of the body. The system features Sinogram Affirmed Iterative Reconstruction (SAFIRE) algorithm for iterative reconstruction to improve the image quality. It also features an Iterative Metal Artifact Reduction (iMAR) algorithm for reducing the metal-related artefact, which occurs due to implants such as dental implants.
RT Pro edition of the system features HD FoV Pro reconstruction, which enhances the visualisation of a more comprehensive CT field of view, improving the image quality for even bariatric patients.
CT device can also be utilised for low-dose screening of lung cancer in high-risk patients.
Other features of Biograph Vision PET / CT system
Biograph Vision PET / CT system features the QualityGuard, Oncofreeze and Cardiofreeze technologies.
QualityGuard utilises the natural radioactive properties of lutetium present in LSO crystals for automatic calibration of the PET detector. The feature reduces staff exposure to radiation by avoiding weekly manual calibration, reducing work-related injury risk and saving up to 30 minutes a day.
Oncofreeze and Cardiofreeze features offer motion-free virtual images of body organs during scanning, improving lesion detection without increasing the scan time.
Technology details
PET / CT system is equipped with artificial intelligence (AI)-enabled AIDAN platform, using proprietary ALPHA technology for scanning operations. The digital hardware of the system processes AI algorithms.
A patient-centric, completely automated FlowMotion™ multi-parametric PET suite is an optional technology integrated into the system. It allows dynamic imaging of the patients and reduces the patient’s exposure time to radiation.
Benefits of Biograph Vision PET / CT system
The system’s wide bore with small tunnel length provides 25% more space than the standard systems. It reduces patient discomfort in small spaces and enables trouble-free positioning for radiotherapy devices and bariatric patients.
Small LSO crystals in the Optiso UDR detector improve spatial resolution and enable better visual details in lower doses for the detection of even small lesions. The system reduces the scan time by a factor of 3.9, avoiding unnecessary exposure to radiation as well as lowering tracer cost.
The detector plate of the system is cooled by high-flow direct-cooling for maintaining the ambient temperature, which avoids the requirement of dehumidifiers, air conditioning and noisy fans.
Biograph Vision’s RT Pro edition allows radiation therapy planning by providing better insights into disease progression for subsequent personalised treatment.